Photoexcitation is recognised as an underused synthesis tool. A brief look at published literature shows that only about 1% of the published papers in the field of organic syntheses involve a photochemical step. However, in photochemistry courses it is taught that because the chemistry of the excited singlet and triplet states are added to that of the ground state excitation by light multiplies by 3 the accessible reaction paths. It therefore appears that photochemical reactions are significantly less used as they could or should be.
It is Vapourtec’s belief that a major reason for the underutilisation of Photoexcitation is the poor suitability of most batch reaction techniques to photochemical reactions. Continuous Photochemical reactors of the falling film or micro-flow tube style are much better tuned to the photon penetration properties of a typical solution of reagent.
Advantages of Continuous Flow Photoexcitation Reactions
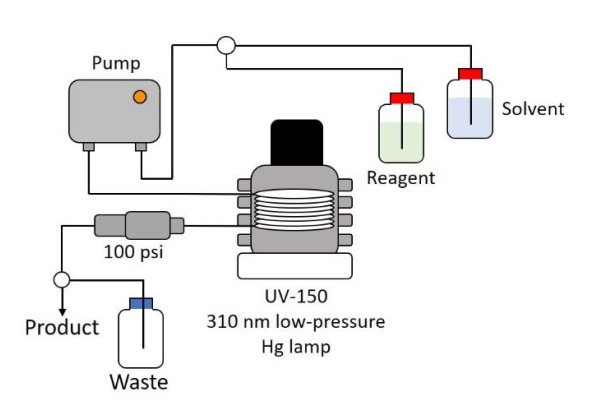
Vapourtec has now made available for both the E-Series and R-Series systems an advanced photochemical reactor intended for continuous flow operation, the UV-150. By eliminating the problems associated with traditional batch photoreactors Vapourtec’s UV-150 enables chemists to take full advantage of the powerful reactions and synthetic routes offered by photo chemical reactions.
The main advantages of flow photoexcitation reactors over conventional batch systems are; consistent light penetrations, controlled exposure times, precise temperature control and removal of the photochemical products from the irradiated area. These features typically result in higher conversions or yields, improved selectivity, enhanced energy efficiencies and reductions of solvent volumes and consequently waste.
Application areas for the Innovative UV-150 Reactor
The UV-150 is a versatile continuous flow photochemical reactor having a broad range of application areas:
- Synthesis of Novel Building Blocks
- Drug Discovery
- Agrochemical Development
- Flavours and Flavourings
- Research into Solar Chemistry (Artificial Photosynthesis)
- Degradation Studies
- UV initiated polymerizations
- Supramolecular Organic Photochemistry