What is hydrogenation?
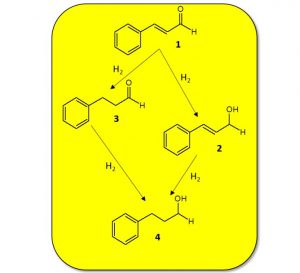
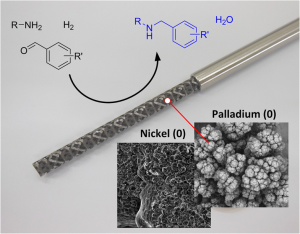
Hydrogenation is the chemical reaction between molecular hydrogen, H2, and another compound or element. There is usually a metal catalyst present, such as palladium, nickel or platinum, which serves to ‘activate’ the hydrogen gas, so it is in a chemically accessible state.
Hydrogenation is typically used to remove double bonds from a molecule through reduction, for example conversion of an alkyne to an alkene, a ketone to an alcohol, or a nitro group to an aniline. Commonly used hydrogenation catalysts include Raney-nickel, palladium on carbon, platinum oxide (Adam’s catalyst), Wilkinson’s catalyst and Lindlar’s catalyst, although the exact catalyst used depends upon the transformation required.
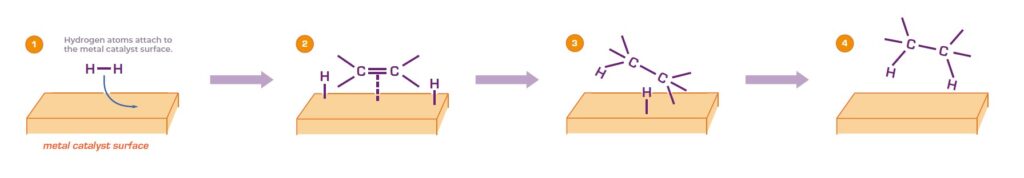
Scheme 1: General mechanism for hydrogenation in an alkene reduction on a metal surface.
Key considerations
The key considerations when undertaking hydrogenation relate to safety, with fire and explosion being primary concerns. In addition, the exothermic nature of the reaction and requirement for flammable solvents, often MeOH or EtOH, poses risks at larger scale [1]. In batch, high-pressure resistant reactors, autoclave conditions and special safety precautions are sometimes necessary, which can all provide barriers to the accessibility and scalability of a transformation [2].
Benefits of continuous flow for hydrogenation
Continuous flow processing can provide excellent control over reagent stoichiometry and temperature leading to a reduction in the risk of side-reactions, as well as the opportunity to re-use catalysts through use of solid supports or cartridges [2, 3, 4]. In addition, there is significant opportunity to improve the safety profile of hydrogenation, particularly upon larger scale: scale-up simply requires passing a stream of substrate through the reactor for longer, rather than using larger equipment [5].
Vapourtec have successfully used both the R-Series and E-Series systems for transfer hydrogenation, hydrogenation using hydrogen gas and a solid catalyst, and coated reactors. Both systems can be easily customised to a particular reaction by varying flow rates, tuning residence times and using a variety of catalysts, both homo- and heterogeneous. Some examples outlining hydrogenation in flow are provided.
Pd/C
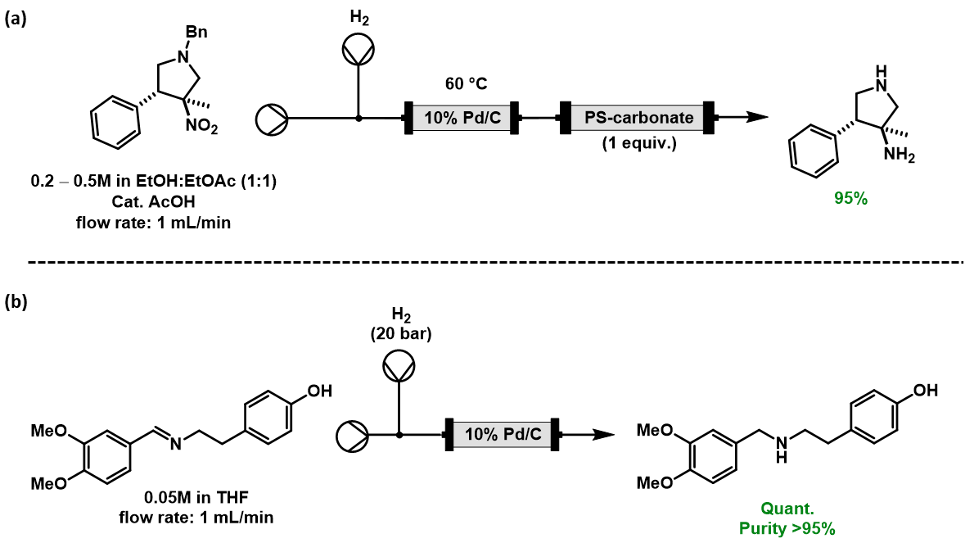
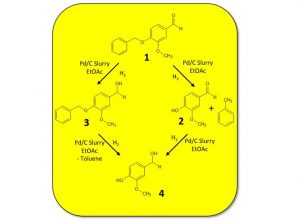
Palladium on carbon (Pd/C) is a common choice as a catalyst for hydrogenation due to its low cost when compared with other transition metal catalysts, the broad range of reductions it can undertake, and its proven ability to reduce heteroaromatic rings [6]. For example, Pd/C can reduce alkenes or alkynes, nitro groups to amines (including anilines) [7], carbonyls to alcohols, and imines to amines [8] as well as effect protecting group removal [3], Scheme 2.
Scheme 2: (a) Reduction of nitro groups to amines with H2 through use of a cartridge containing Pd/C and in-line purification [7]; (b) Reduction of imines to amines with H2 using Pd/C loaded into a cartridge [8].
Pd/C slurries in flow
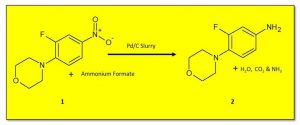
When using Pd/C in batch mode, a slurry of the catalyst and reagent is prepared, usually in MeOH or EtOH, and this mixture is then exposed to hydrogen gas. While handing slurries and heterogeneous materials is commonplace within batch chemistry, under flow mode slurries can lead to issues with blockages. To circumvent this, catalysts such as Pd/C can be packed into a cartridge, although this method can be scale-limited due to back-pressure build-up when pumping though a filled column, as well as catalyst degradation over time.
The Vapourtec easy-MedChem can be used to pump suspensions and slurries with particles of up to 80 μm in diameter, with no blockages or pressure build-up observed. Using this set-up, both reduction of a nitro group to an aniline with Pd/C using transfer hydrogenation conditions, as well as the reduction of O-benzyl vanillin using hydrogen gas, have been achieved.
Lindlar’s Catalyst
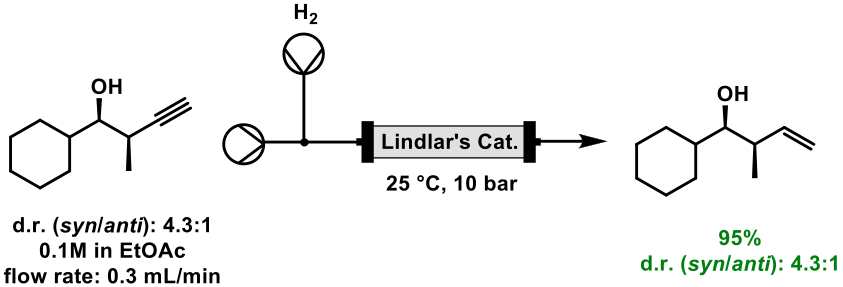
Lindlar’s catalyst, also known as ‘poisoned palladium’, consists of palladium deposited on calcium carbonate or barium sulfate, that is then poisoned with lead or sulfur. It is specifically used for reduction of alkynes to alkenes, without over-reduction to alkanes, and has been exploited within flow chemistry to reduce a terminal alkyne to an alkene, with no erosion of diastereomeric ratio [9], Scheme 3.
Scheme 3: Reduction of a terminal alkyne to alkene in flow mode with Lindlar’s catalyst.
Wilkinson’s catalyst
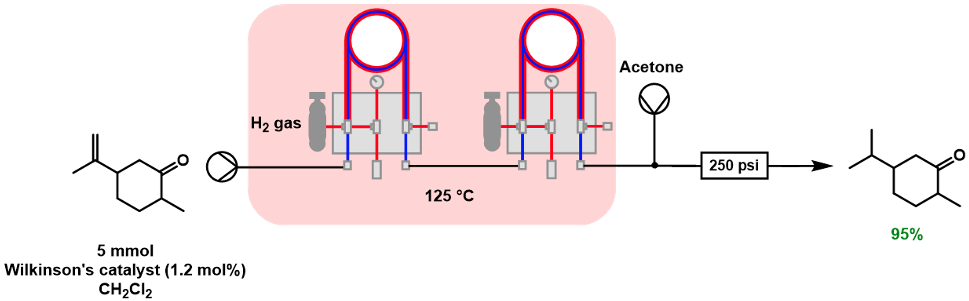
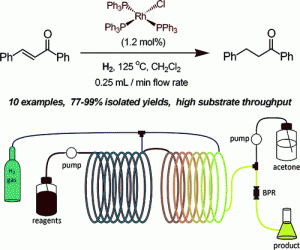
In the previous examples, heterogeneous catalytic systems were considered. However, use of homogeneous catalysts is also possible, although purification to remove the catalyst (either in-line or as an added operation) is required. Leadbeater and co-workers used Wilkinson’s catalyst to undertake a series of alkene reductions with the Vapourtec gas-liquid reactor, which also allowed heating to 125°C [10]. Purification involved passing the crude material through a silica plug to remove any impurities, Scheme 4.
Scheme 4: Use of Wilkinson’s catalyst to reduce an alkene to a methylene.
Asymmetric reduction
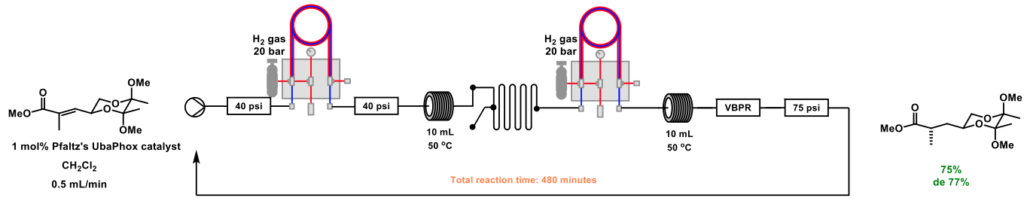
Asymmetric reduction has also been achieved using continuous flow processing. Screening of catalytic conditions was expediated using a flow chemistry set-up, and several tri-substituted olefins were successfully treated with catalysts based on either iridium, ruthenium or rhodium in conjunction with hydrogen gas, using a tube-in-tube reactor [11]. Recirculation of material allowed low catalyst loadings (1 mol%) to be used, with no depreciation in diastereometeric ratio observed, even after a total reaction time of 480 minutes, Scheme 5.
Scheme 5: Asymmetric reduction in flow. Recirculation of material allowed low catalyst loadings to be used, with no detrimental effect on diastereoselectivity.
Biocatalysis
Biocatalysis is a useful alternative to traditional metal-mediated catalysis due to the high chemo-, regio- and stereoselectivity that allows for efficient production of enantiopure materials through use of enzymes, which often require milder conditions than metal-mediated transformations. However, there are key challenges with their use including the high cost and limited stability of the catalyst, as well as the requirement of many redox enzymes for expensive co-factors [12]. Continuous flow chemistry is actively being explored for biocatalysis, with advantages including increased productivity, improved safety profiles, automation, in-line monitoring and the possibility for catalyst recovery and re-use. Under continuous flow processing, biocatalysis has been successfully used to enantioselectivity reduce ketones to the corresponding alcohols in excellent yields and enantiopurity, as well as opening the possibility for selective deuterium labelling [12].
References
[1] Hazards associated with laboratory scale hydrogenations (T. Chandra and J. P. Zebrowski, J. Chem. Health Saf., 2016, 23, 4, 16 – 25) https://doi.org/10.1016/j.jchas.2015.10.019
[2] Heterogeneous Catalytic Hydrogenation Reactions in Continuous-Flow Reactors (M. Irfan, T. N. Glasnov, C. O. Kappe, Chem. Sus. Chem., 2011, 4, 300 – 316) https://doi.org/10.1002/cssc.201000354
[3] The expanding utility of continuous flow hydrogenation (P. J. Cossar, L. Hizartzidis, M. I. Simone, A. McCluskey and C. P. Gordon, Org. Biomol. Chem., 2015, 13, 7119 – 7130) https://doi.org/10.1039/c5ob01067e
[4] Recent Progress in Continuous-Flow Hydrogenation (T. Yu, J. Jiao, P. Song, W. Nie, C. Yi, Q. Zhang and P. Li, Chem. Sus. Chem., 2020, 13, 2876 – 2893) https://doi.org/10.1002/cssc.202000778
[5] The Use of Gases in Flow Synthesis (C. J. Mallia, I. R. Baxendale, Org. Proc. Res. Dev., 2016, 20, 327 – 360) https://doi.org/10.1021/acs.oprd.5b00222
[6] A Continuous Flow Strategy for the Facile Synthesis and Elaboration of Semi-Saturated Heterobicyclic Fragments (N. Luise, E. W. Wyatt, G. J. Tarver and P. G. Wyatt, Eur. J. Org. Chem., 2019, 1341 – 1349) https://doi.org/10.1002/ejoc.201801684
[7] Synthesis of 3-Nitropyrrolidines via Dipolar Cycloaddition Reactions Using a Modular Flow Reactor (M. Baumann, I. R. Baxendale, S. V. Ley, Synlett, 2010, 5, 749 – 752) https://doi.org/10.1055/s-0029-1219344
[8] The use of a continuous flow-reactor employing a mixed hydrogen–liquid flow stream for the efficient reduction of imines to amines (S. Saaby, K. R. Knudsen, M. Ladlow, S. V. Ley, Chem. Commun., 2005, 2909 – 2911) https://doi.org/10.1039/B504854K
[9] Diastereoselective Chain-Elongation Reactions Using Microreactors for Applications in Complex Molecule Assembly (C. F. Carter, H. Lange, D. Sakai, I. R. Baxendale, S. V. Ley, Chem. Eur. J., 2011, 17, 3398 – 3405) http://dx.doi.org/10.1002/chem.201003148
[10] Continuous Flow Hydrogenation Using an On-Demand Gas Delivery Reactor (M. A. Mercadante, C. B. Kelly, C. (X.) Lee and N. E. Leadbeater, Org. Proc. Res. Dev., 2012, 16, 5, 1064 – 1068) https://dx.doi.org/10.1021/op300019w
[11] Asymmetric Homogeneous Hydrogenation in Flow using a Tube-in-Tube Reactor (S. Newton, S. V. Ley, E. C. Arcé and D. M. Grainger, Adv. Synth. Cat., 2012, 354, 1805 – 1812) https://doi.org/10.1002/adsc.201200073
[12] Rapid, Heterogeneous Biocatalytic Hydrogenation and Deuteration in a Continuous Flow Reactor (L. A. Thompson, J. S. Rowbotham, J. H. Nicholson, M. A. Ramirez, C. Zor, H. A. Reeve, N. Grobert and K. A. Vincent, Chem. Cat. Chem., 2020, 12, 3913 – 3918) https://doi.org/10.1002/cctc.202000161