What is Oligonucleotide synthesis?
Oligonucleotide synthesis is a chemical process used to create short sequences of nucleotides, which are the building blocks of DNA and RNA. This technique enables the production of precise and custom-designed oligonucleotides for a variety of applications in molecular biology, medicine, and biotechnology. The synthesis typically involves the step-by-step addition of nucleotide units to a growing chain, using a solid-phase method that ensures high efficiency and fidelity. This process has revolutionized fields such as genetic testing, and therapeutic development.
Oligonucleotide synthesis in modern drug discovery
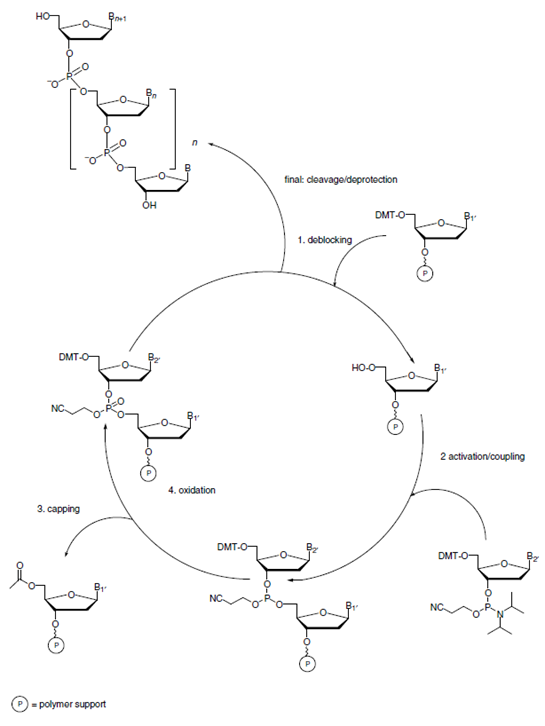
Synthesis of oligonucleotides, such as DNA or RNA molecules, using solid-phase methods has been of immense importance to developing modern genetics, cell and molecular biology and drug discovery. The development of solid phase synthesis methods for oligonucleotides started almost simultaneously alongside peptide synthesis in the 1960’s and 70’s and became very efficient when Caruthers et al. introduced phosphoramidite methodology in 1980’s. In fact, phosphoramidite chemistry has been so successful that very little has changed in oligonucleotide synthesis since its introduction. The original applications for synthetic oligonucleotides, such as DNA sequencing or PCR primers, needed these products in only sub-micromole scale. Later, relatively larger quantities of these molecules were needed for antisense gene expression control, diagnostic assays, and physical studies of nucleic acids. In the last decade or so the demand on oligonucleotides and their production has increased significantly due to genetic medicines coming of age. Various types of medicines have either been marketed or are under early or advanced clinical investigation. These include gene therapy, RNA medicines (e.g. vaccines), gene editing (e.g. CRISPR-Cas9) and RNAi therapeutics. While the first three groups are currently produced using molecular biology techniques, the last group, RNAi therapeutics are made via total chemical synthesis. Although from a drug discovery point of view these agents are still facing major obstacles such as addressing delivery challenges, it is fair to say that their production on large scale can be considered as one of the main bottlenecks to their commercialization. This challenge becomes more evident when we consider gradual change of target diseases from rare to more prevalent diseases. For example, while drugs used for treating hereditary transthyretin amyloidosis (hATTR) might be needed on kilogram scale annually, those used for hypertension such as zilebesiran will be needed on ton scale if they can successfully penetrate these very large markets. As such there is a significant need for increased RNA manufacturing capacity.
Existing technology challenges in oligonucleotide synthesis
The current technology used for synthesis of these RNAi targets, although mature and efficient is only suitable for addressing the demand for rare diseases. The synthesis of a typical 21-mer SiRNA duplex involves more than 160 steps and yields of 50% and purity in the region of 90% are considered a significant achievement. Currently a typical API costs more than $1m per kg. This is due to expensive raw material but also process mass intensity (on average 5 tons of raw material to produce 1kg of API), expensive equipment and lengthy process and most importantly low yield and efficiency. There are also other concerns, such as difficult scalability of the process, environmental impact of very high raw material consumption, inconsistent batch quality and low overall purity and yield of the final API. As a result, a new process that can improve any of these constraints is highly welcome and needed by the industry.
Flow chemistry offers improvements in oligonucleotide synthesis
A key challenge in the large-scale synthesis of oligonucleotides is the support on which the synthesis is performed. Ideally a support should be physically robust to allow mixing to increase reaction efficiency without damaging the matrix and so controlled pore class (CPG) or macroporous resins are used. However, these have low loadings and large quantities are needed for a scaled-up synthesis which leads to heat and mass transfer challenges. To increase capacity, supports have been developed with a large surface area but these materials have inherent porosity that leads to irreversible entrapment of reagents in the matrix. These polymers, when used in a conventional mixed bed reactors or the currently used static compression technology, trap the reagents in their pores and require high volumes of solvent to remove the entrapped reagents. When using very large amounts of resin, even use of significant volumes of solvent may not be enough to displace the reagents.
This trade-off between the surface and porosity has led researchers to hunt for a perfect polymeric support by studying the nature of the polymer, cross-linking level, porosity and other chemical or physical properties. The variable-bed flow reactor (VBFR) technology developed by Vapourtec, enables a different approach to the problem. This patented reactor ensures a perfect packed bed reactor by maintaining the resin under a constant density of packing by adjusting the reactor volume accordingly.
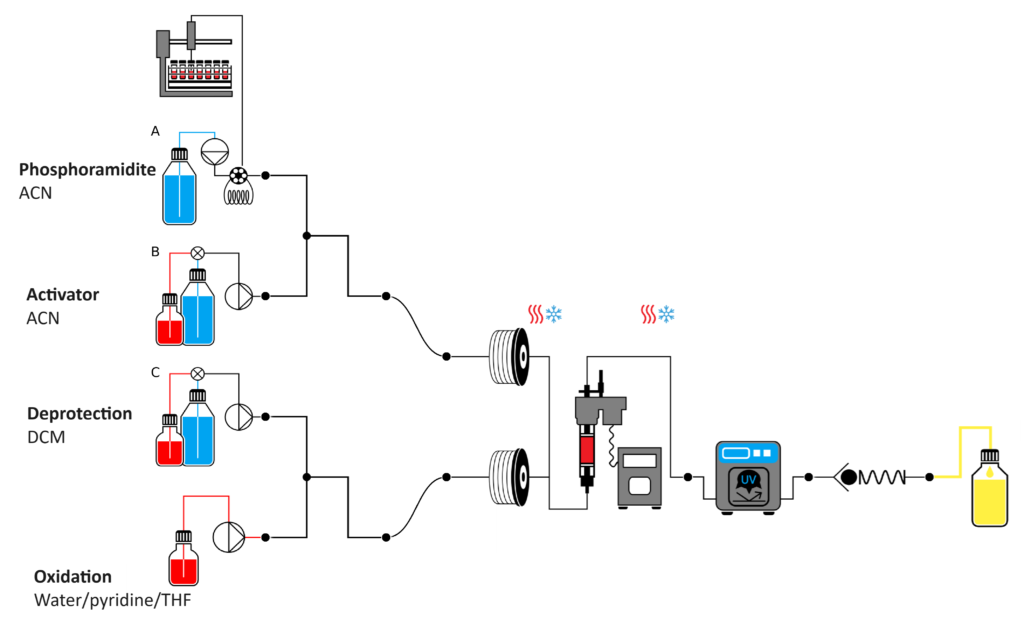
Figure 2. Typical configuration for single pass oligonucleotide synthesis using a Vapourtec synthesizer
Using VBFR technology allows augmented diffusivity by convection, instead of relying on diffusion. This convective flow effectively “pushes out” the reagents from the pores resulting in the use of much smaller amounts of solvent in wash steps. Also, by using the high-capacity polymers the detrimental effect of residual water on the coupling step would be reduced hence allowing us to use lower equivalents of the nucleotides and activators in the synthesis. Finally, the flow configuration gives a better control of mass and heat transfer, leading to a more efficient synthesis protocol. For example, in a flow reactor the residence time of deprotecting reagent can be adjusted precisely in a way that all protecting groups are removed but the acid-induced depurination side reaction is avoided.