Flow chemistry is also known as continuous flow or plug flow chemistry. It involves a chemical reaction run in a continuous flow stream. The process offers the potential for the efficient manufacture of chemical products. Significant breakthroughs using flow chemistry systems are in the production of Tamoxifen (Breast Cancer) and Artemisinin (Malaria).
Reactants are first pumped into a mixing device. Flow continues through a temperature controlled reactor until the reaction is complete. The reactor can be a simple pipe/tube or a complex micro-structured device. The mixing device and reactor are maintained at the precise temperature to promote the desired reaction. The reactants may also be exposed to an electrical flux or a photon flux to promote an electrochemical or photochemical reaction.
Click here for examples of chemistry performed in Vapourtec systems
Flow chemistry differs from conventional batch chemistry by having the following important features:
Flow of reagents
In Flow, chemistry reagents are pumped under pressure and flow continuously through the reactor. This contrasts with batch reactors where all reagents are loaded into a vessel at the start
Control of reaction time
Reaction time is determined by the time the reagents take to flow through the reactor. This period is called the residence time.
Control of stoichiometry
Reaction stoichiometry is controlled by the relative flow rates of the reactants. The concentration of one reagent relative to another can be increased simply by pumping that reagent at a higher flow rate.
Heat transfer
Flow reactors have excellent heat transfer when compared with batch reactors. This is because the surface area to volume ratio of flow reactors is much greater than that of batch reactors.
Mass transfer
Reactors designed for flow chemistry have high rates of mass transfer. This is due to the small sizes and good mixing that is possible.
Flow chemistry is easily scaled
Flow reactions can simply be run for longer. This produces more material.
Precise control
Flow offers the chemist precise control of the four critical reaction parameters. These parameters are stoichiometry, mixing, temperature, and reaction time
Low inventory of materials
When reactions are run in continuous flow only small quantities of potentially hazardous materials are “in-process”.
Telescoped reactions
Reactive intermediates don’t need to be isolated. Flow reactions can be easily run in sequence or “telescoped”.
No head-space
Flow reactors do not require a head space. The pressure within the reactor is controlled by a back pressure regulator (BPR). With high pressure batch reactors, the gas within the headspace must be pressurised.
Very low back-mixing
Flow reactors can be arranged to have very little or even no back-mixing
Click here for examples of chemistry performed in continuous flow.
Examples of flow chemistry available from International publications:
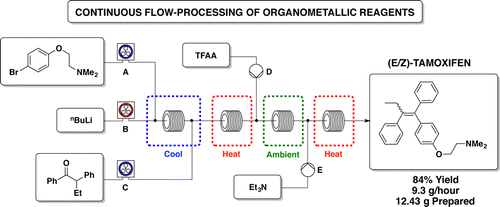
Continuous Flow-Processing of Organometallic Reagents Using an Advanced Peristaltic Pumping System and the Telescoped Flow Synthesis of (E/Z)- Tamoxifen
- Philip R D Murray
- Duncan L Browne
- Julio C Pastre
- Chris Butters
- Duncan Guthrie
- Steven V Ley
- Dept. of Chemistry, University of Cambridge – UK Instituto de Química, University of Campinas, Brazil – Vapourtec Ltd, UK
This paper describes several examples of the use of organometallic reagents in a Vapourtec flow chemistry system. These include n-butyllithium, Grignard reagents, and DIBAL-H. Examples are reported over several hours of continuous pumping. Multigram quantities of products are produced. The highlight of the paper is an approach to the telescoped synthesis of (E/Z)-tamoxifen. Organometallic reagent-mediated transformations are used.
Click here to go to the full ACS Publication
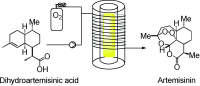
Continuous-Flow Synthesis of the Anti-Malaria Drug Artemisinin
- François Lévesque
- Peter H. Seeberger
- Department for Biomolecular Systems, Max-Planck Institute for Colloids and Interfaces, Germany – Institute for Chemistry and Biochemistry, Freie Universität Berlin, Germany
This paper describes the use of flow chemistry techniques to develop a continuous-flow process. The process converts dihydroartemisinic acid into artemisinin in an inexpensive, scalable process. Artemisinin combination treatments are the first-line drugs in the global fight against Malaria.
Click here to go to the full Angewandte Chemie Publication
Other flow chemistry resources:
For an overview of flow chemistry advantages, parameters, and reactors by Dr. Claudio Battilocchio and Prof Steven V. Ley (Innovative Technology Centre, University of Cambridge, United Kingdom) please visit the article link below:
To see the organic chemistry portal flow chemistry article click here
For further information on the subject of flow chemistry at nature.com please visit the article using the link below: