When applied to chemical plants, Process Intensification (PI) is the strategy of making dramatic improvements in manufacturing and processing, substantially decreasing, equipment size to output ratio, waste production, and energy consumption. These changes ultimately result in sustainable chemical plants that are cheaper to operate.
From an engineering standpoint, Process Intensification is any chemical engineering development that leads to significantly smaller, cleaner, and more energy efficient manufacturing plant.
History of process intensification
During the late 1970’s the UK based Imperial Chemical Industries (ICI) pioneered the concept of process intensification. ICI aimed to reduce the size of new chemical plants to majorly decrease the capital costs of new plants. The philosophy of ICI was based on the premise that the majority (up to 80%) of the plant cost was attributable to installation costs involving basics like pipework, support structure, and civil engineering. These costs could be significantly reduced if the main, “active” plant items (i.e. reactors, separators, and heat exchangers) were significantly reduced in size and/or coupled together by integrating equipment function. To access these cost reductions ICI established a programme of process intensification.
Process intensification today
Today the focus on process intensification has moved forwards from the late 1970s ICI model of plant size/cost reduction. Process intensification is now re-focused on creating a competitive advantage through not only plant size/cost reduction but also via improved quality, higher total process efficiency, and increased operational flexibility.
Many “intensified” chemical manufacturing processes have been developed and processing equipment engineered. A large proportion of these solutions are based around the concepts of continuous flow processing as opposed to batch processing. Continuous flow reaction processes are normally known collectively as flow chemistry. The goals of process intensification can be achieved by either using chemical reactions that cannot be conducted in larger volumes or by running chemical reactions at parameters (temperatures, pressures, concentrations) that are inaccessible in larger volumes due to safety constraints.
For example, the detonation of the stoichiometric mixture of two volumes of hydrogen and one volume of oxygen gas will not propagate in microchannels with a sufficiently small diameter. This is an example whereby “intrinsic safety” can be built into a process by using microstructured reactors. In many cases, the yields and selectivity of reactions have been improved by using novel reactions or running reactions at more extreme conditions.
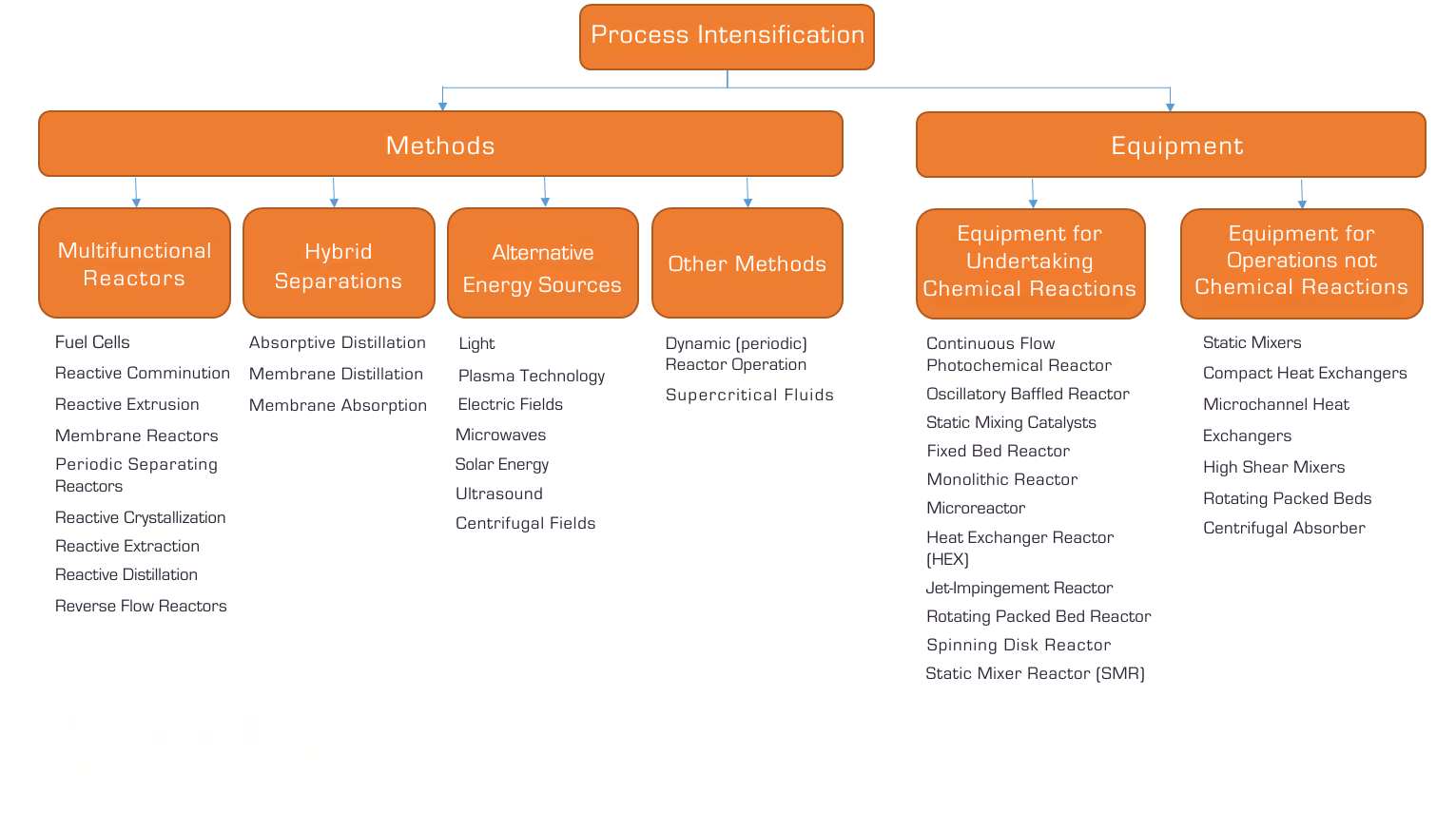
Many of the technologies used in process intensification are shown in the chart below:
Examples of process intensification available from international publications:
Spinning disk reactor for the intensification of styrene polymerisation
- K.V.K. Boodhoo
- R. J. Jachuck
In this paper, the performance of a thin film reactor using the spinning disc technology has been described. The objective of the study was to develop a compact reactor with good heat and mass transfer characteristics for continuous processing of free radical polymerisation. Solution polymerisation of styrene was therefore chosen as an example of a free-radical polymerisation system and it may be concluded that a spinning disc reactor (SDR) can significantly enhance the reaction rates when the viscosity of the polymer mass is more than 3–4 N/m2 s. It also improves product quality by producing polystyrene with tight molecular weight distribution (MWD). The inherent safety of the process is also improved.
Click here to go to the full ATE Publication
Development of a continuous flow scale-up approach to reflux inhibitor AZD6906
- Tomas Gustafsson
- Henrik Sörensen
- Fritiof Pontén
Early scale-up work of a promising reflux inhibitor AZD6906 is described. Two steps of an earlier route were adapted to be performed in a continuous flow to avoid issues related to batch procedures, resulting in a robust method with reduced cost of goods and improved product quality. Toxic and reactive reagents and starting materials could be handled in a flow regime, thereby allowing safer and more convenient reaction optimization and production.