Enzymatic reactions in flow: a new opportunity for sustainability, efficiency and productivity
Biocatalysis specialises in the use of enzymes to perform chemical transformations – enzymatic reactions – and is often promoted as being cheaper, cleaner and more environmentally friendly than ‘traditional’ organic chemistry. The use of enzymes for synthesis (enzymatic reactions) can provide opportunities to improve reaction efficiency and productivity, as well as address sustainability. In addition, precise control of a transformation is possible, enabling outstanding stereo-, regio-, and chemoselectivity [1].
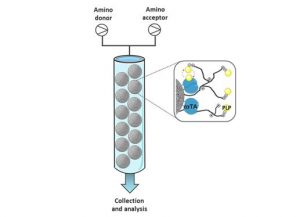
Although the use of bioreactions within mainstream organic chemistry is established, translation into flow chemistry has been slower. However, in recent years there have been significant advances in this field. In particular, enzyme immobilisation has allowed the development of column or ‘packed-bed’ reactors [2, 3].
Flow reactors from Vapourtec have been used extensively by researchers looking to exploit the use of enzymatic reactions, with the modularity of the R-Series providing significant advantages. This short article will highlight recently disclosed biotransformations, where using flow mode often achieves higher yields, faster reaction rates, and better selectivity, as well as increased retention of enzyme activity when compared to the batch process.
Biocatalysis using Candida antarctica (Novozyme 435®)
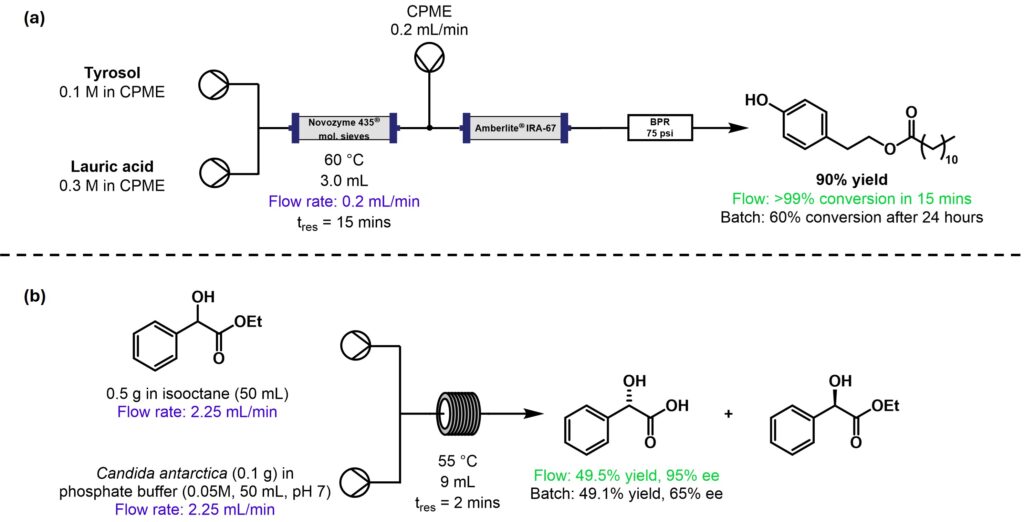
Candida antarctica (Novozyme 435®) is a commercially available immobilised lipase that is stable under a range of solvent conditions. The immobilised form can be loaded into a glass packed-bed reactor column, which fits modularly within the R-Series system. In the two examples showcased, biocatalysis in flow gave superior yields when compared with batch mode, and in one example an in-line work-up streamlined the purification process to simple solvent evaporation, Scheme 1.
Scheme 1: (a) Generation of phenolic esters as antiradical and antimicrobial agents [4]; (b) Kinetic resolution of chiral mandelic acids [5]. NB the highest yield possible for this reaction is 50%; stereochemical configuration of products taken from original schemes.
Biocatalysis using immobilised acyl transferase from Mycobacterium smegmatis
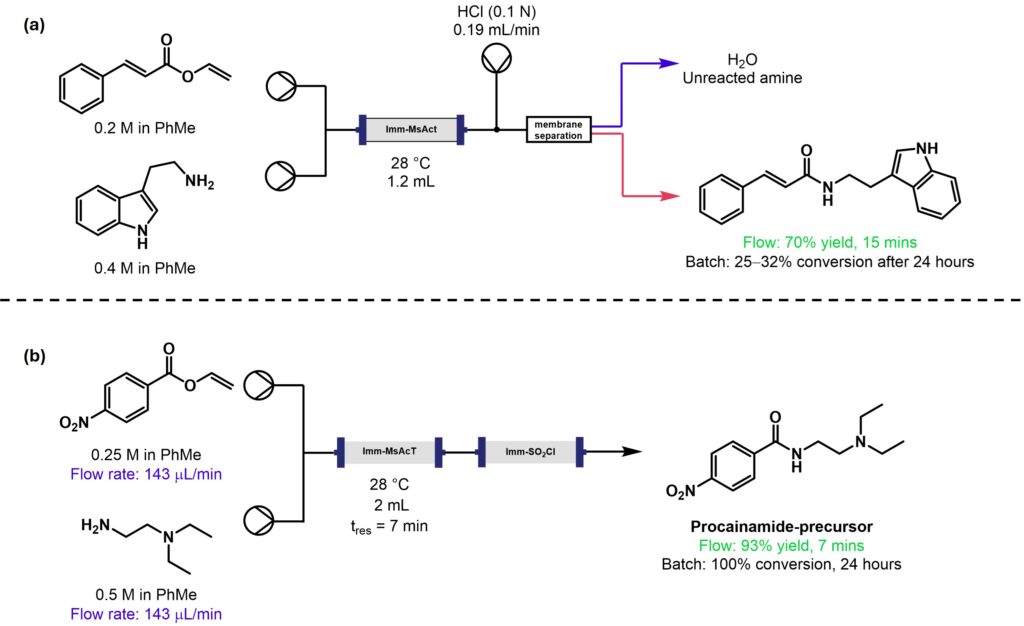
In this example, acyltransferase from Mycobacterium smegmatis was immobilised onto either glyoxyl-agarose or glyoxyl-garose beads, allowing easy incorporation into the Vapourtec R-Series through use of a glass packed-bed reactor column, Scheme 2. Residence time could be adjusted by variation of flow rate, ensuring complete conversion of the reactants. In example (a), the use of flow led to fewer by-products and higher yields.
Scheme 2: (a) Multi-gram preparation of cinnamoyl tryptamines [6]; (b) Enzymatic synthesis of a precursor to procainamide and other APIs [7].
Enzymatic reactions in the synthesis of the antiviral vidarabine
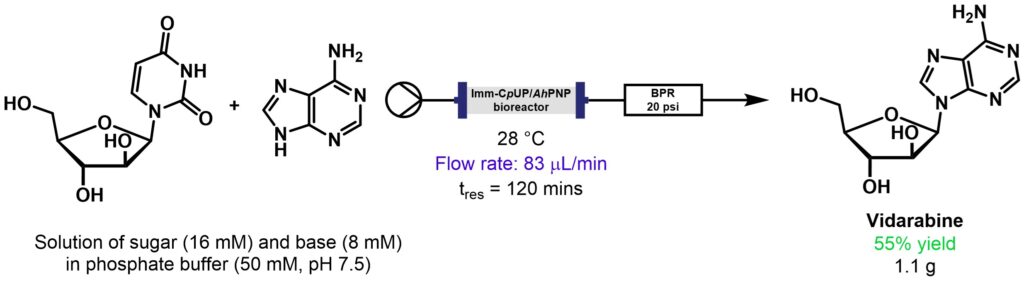
Vidarabine is an antiviral drug active against herpes simplex and varicella zoster viruses. This biotransformation was catalysed through a combination of uridine phosphorylase from Clostridium perfringens (CpUP) and purine nucleoside phosphorylase from Aeromonas hydrophila (AhPNP). As well as undertaking the desired transformation in a Vapourtec R-Series system, the set-up was also used to immobilise the enzymes by pre-packing the hydrophilic supports into a glass packed-bed reactor column and flowing a solution of the enzyme through, Scheme 3. HPLC analysis showed that the enzymes in flow-mode retained their activity even after 5 days continuous work.
Scheme 3: Synthesis of the antiviral vidarabine using immobilised CpUP and AhPNP [8].
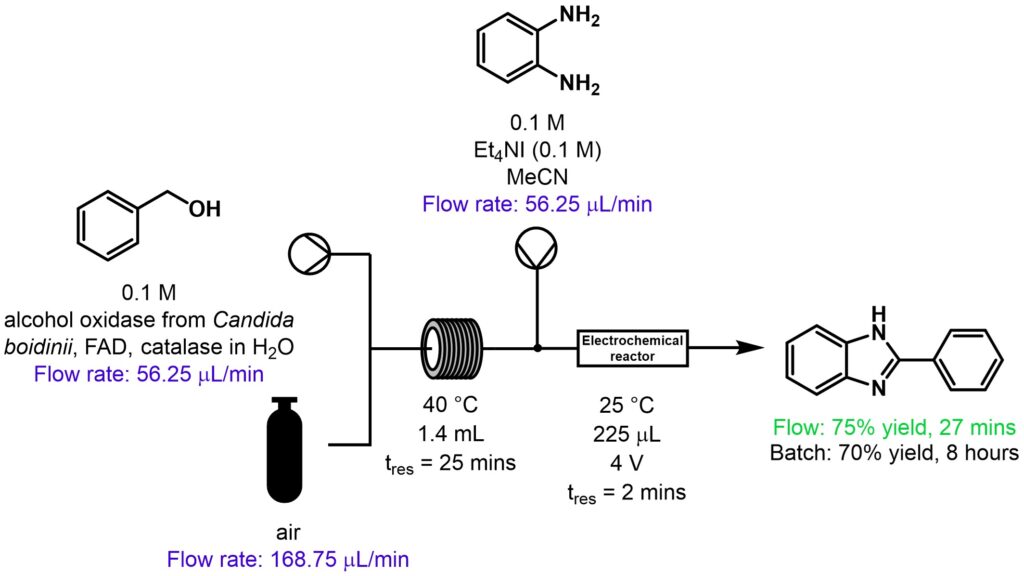
Biotransformations using alcohol oxidase from Candida boidinii, FAD and a catalase then electrochemical cyclisation
Substituted benzimidazoles can exhibit numerous useful properties, including antiparasitic, antiallergic, analgesic, anti-inflammatory, antidepressant and anticancer activity. In this example, a range of benzimidazoles was prepared using a two-step telescoped process including biocatalysis and electrochemical cyclisation, Scheme 4 [9].
Scheme 4: Enzymatic synthesis of a range of benzimidazoles [9].
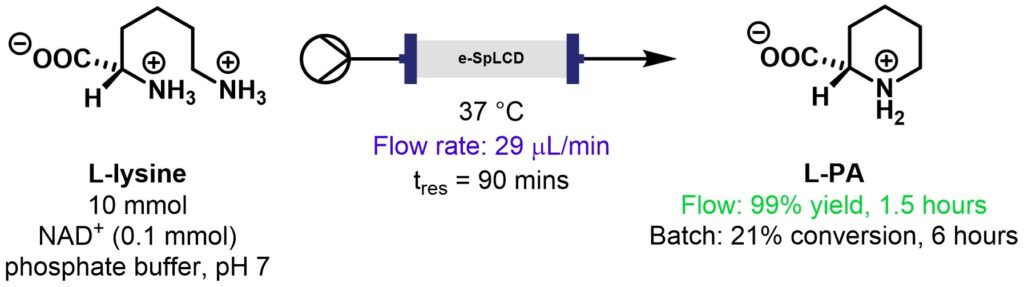
Biocatalytic synthesis of L-pipecolic acid by a lysine cyclodeaminase from Streptomyces pristinaespiralis
The synthesis of non-proteinogenic amino acids (NPAAs) is gaining attention due to the novel bioactive properties of this class of compound. The Paradisi group prepared L-PA cheaply and efficiently from L-lysine using enzymatic synthesis, avoiding the harsh conditions and hazardous reagents used in previous approaches, Scheme 5 [10]. The use of flow resulted in much shorter reaction times and excellent yields. In addition, the immobilised enzyme fully retained activity after 120 hours at 37 °C whereas the free enzyme lost 70% of initial activity after 96 hours.
Scheme 5: Synthesis of L-PA from L-lysine [10].
In summary, enzyme-mediated synthesis using flow chemistry offers key advantages when compared with the ‘traditional’ batch approach. For example, yields and conversion rates are often better, and the enzyme itself can exhibit higher biocatalytic productivity. The Vapourtec R-Series has been successfully used for a range of biotransformations, either through passing a mixture of enzyme and substrate through a heated coil, or by using immobilised enzymes loaded into a glass packed-bed reactor column.
Contact us today to find out how Vapourtec can support your research.
References
[1] Power of Biocatalysis for Organic Synthesis (C. K. Winkler, J. H. Schrittwieser, and W. Kroutil, ACS Cent. Sci., 2021, 7, (1), 55–71). https://doi.org/10.1021/acscentsci.0c01496
[2] Flow biocatalysis 101: design, development and applications (A. I. Benítez-Mateos, M. L. Contente, D. R. Padrosa and F. Paradisi, React. Chem. Eng., 2021, 6, 599–611). https://doi.org/10.1039/D0RE00483A
[3] The joint effort of enzyme technology and flow chemistry to bring biocatalytic processes to the next level of sustainability, efficiency and productivity (S. Donzella and M. L. Contente, J. Flow Chem., 2023, 14 (85), 85–96). https://doi.org/10.1007/s41981-023-00286-w
[4] Enzymatic continuous-flow preparation of nature-inspired phenolic esters as antiradical and antimicrobial agents (F. Annunziata, M. L. Contente, V. Anzi, S. Donzella, P. Conti, F. Molinari, P. A. Martino, G. Meroni, V. M. Sora, L. Tamborini and A. Pinto, Food Chemistry, 2022, 390, 133195). https://doi.org/10.1016/j.foodchem.2022.133195
[5] Enzymatic kinetic resolution in flow for chiral mandelic acids (S. Wang, T. Shi, Z. Fang, C. Liu, W. He, N. Zhu, Y. Hu, X. Li and K. Guo, Journal of Flow Chemistry, 2022, 12, 227–235). https://doi.org/10.1007/s41981-022-00219-z
[6] Multi-gram preparation of cinnamoyl tryptamines as skin whitening agents through a chemo-enzymatic flow process (D. R. Padrosa and M. L. Contente, Tetrahedron Letters, 2021, 86, 153453). https://doi.org/10.1016/j.tetlet.2021.153453
[7] Efficient Chemo-Enzymatic Flow Synthesis of High Value Amides and Esters (F. Annunziata, M. L. Contente, D. Betti, C. Pinna, F. Molinari, L. Tamborini and A. Pinto, Catalysts, 2020, 10, 939). https://doi.org/10.3390/catal10080939
[8] An Enzymatic Flow-Based Preparative Route to Vidarabine (L. Tamborini, C. Previtali, F. Annunziata, T. Bavaro, M. Terreni, E. Calleri, F. Rinaldi, A. Pinto, G. Speranza, D. Ubiali, and P. Conti, Molecules 2020, 25, 1223). https://doi.org/10.3390/molecules25051223
[9] Enzymatic electrochemical continuous flow cascade synthesis of substituted benzimidazoles (T.-T. Shi, S.-Z. Wang, Z. Yang, Y. Wang, C. Liu, W. He, Z. Fang and K. Guo, React. Chem. Eng., 2021, 6, 937). https://doi.org/10.1039/D1RE00058F
[10] Biocatalytic Synthesis of L-Pipecolic Acid by a Lysine Cyclodeaminase: Batch and Flow Reactors (K. Stalder, A. I. Benítez-Mateos, and F. Paradisi, ChemCatChem 2024, e202301671). https://doi.org/10.1002/cctc.202301671