Synthesis of Cycloalkyl Substituted 7-Azaindoles via Photoredox Nickel Dual Catalytic Cross-Coupling in Batch and Continuous Flow
- Natalie Palaychuk, Travis J. DeLano, Michael J. Boyd, Jeremy Green, and Upul K. Bandarage
- Vertex Pharmaceuticals Incorporated, 50 Northern Avenue, Boston, Massachusetts 02210, United States
 Read the publication that featured this abstract
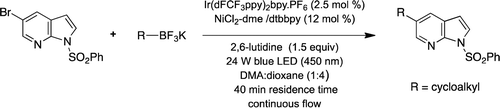
Read the publication that featured this abstractAn efficient photoredox/Ni dual catalytic Csp2–Csp3 cross-coupling protocol in a continuous-flow regime to synthesize a variety of regioisomeric cycloalkyl substituted 7-azaindoles has been developed. These transformations proceed efficiently under mild conditions (blue LED light irradiation at 30 °C over 40 min residence time in mixed solvent systems). Reactions are easy to perform and afford most of the desired 2-, 3-, 4-, 5-, and 6-cycloalkyl substituted 7-azaindoles in moderate-to-good yield.
Get in touch
For more information on flow chemistry systems and services please use the contact methods below.
Call us on +44 (0)1284 728659 or Email us
Resource Centre
R-Series

The Vapourtec R-Series is, quite simply, unrivalled for flow chemistry
- Flexible |
- Precise |
- Automatable
The R-Series is undoubtedly the most versatile, modular flow chemistry system available today.
E-Series

The Vapourtec E-Series is the perfect introductory system for flow chemistry
- Robust |
- Easy to use |
- Affordable
The E-Series is a robust and affordable, entry level flow chemistry system designed for reliability and ease of use.
