What is Green Chemistry?
Green chemistry is a way of doing chemistry that focuses on making it safer and better for the environment. Instead of using traditional methods that create harmful waste, green chemistry works to reduce pollution, save energy, and use materials that are renewable and less toxic. By starting with safer materials and processes, green chemistry helps prevent problems before they happen. This approach is especially important now, as we face challenges like climate change and pollution. The twelve principles of green chemistry guide this work, offering clear steps to create safer and cleaner chemical products.
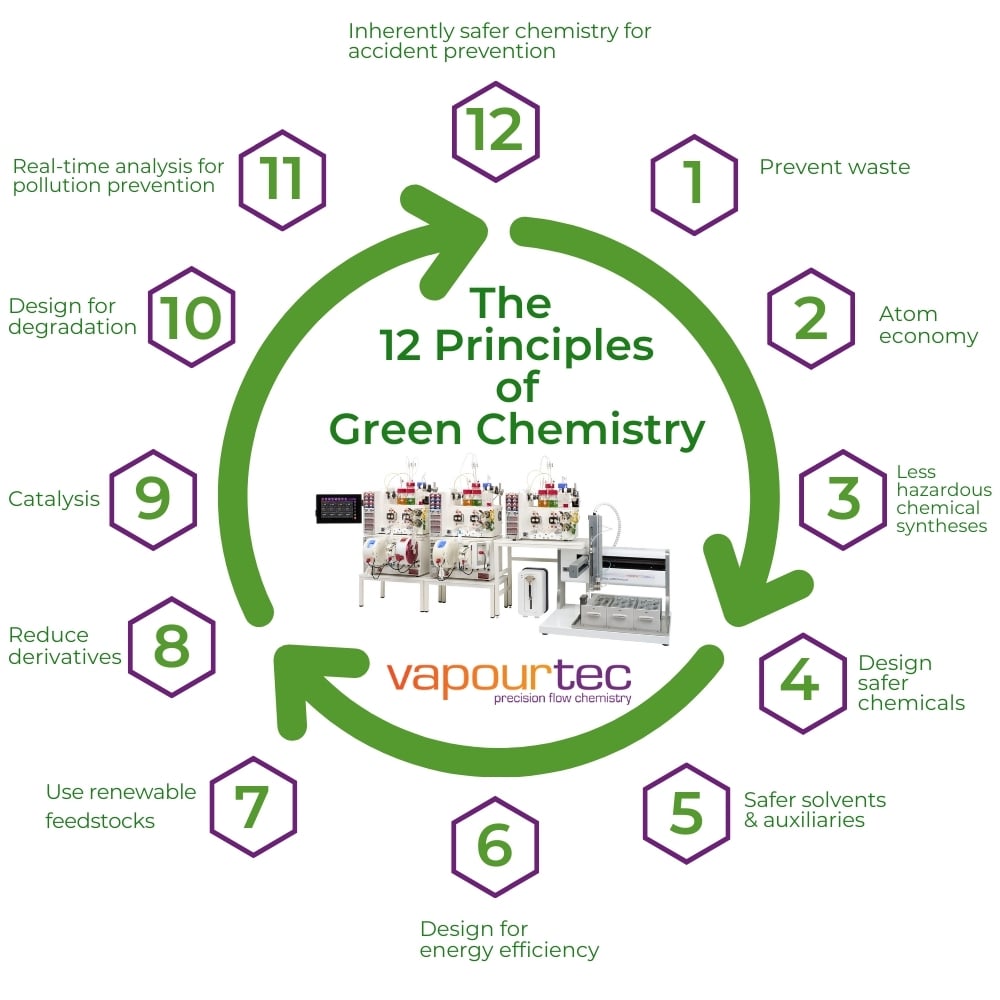
12 Principles of Green Chemistry
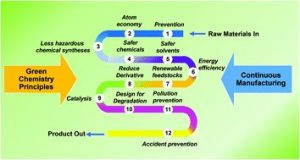
The 12 principles of green chemistry cover every stage of a chemical product’s life, from how it’s made to how it’s disposed of. These principles help chemists design products and processes that are less harmful to people and the planet.
Paul Anastas and John Warner introduced the 12 principles in 1998 to help guide safer and greener practices in chemistry. These principles are:
Prevention:
Stop waste from being created instead of dealing with it later.
Atom Economy:
Use materials in a way that ensures most of them end up in the final product.
Less Hazardous Chemical Syntheses:
Use and make chemicals that are safer for people and the environment.
Designing Safer Chemicals:
Make products that work well while being less toxic.
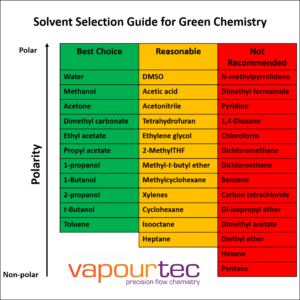
Safer Solvents and Auxiliaries:
Avoid using extra substances like solvents when possible, or choose safer options.
Design for Energy Efficiency:
Use less energy by doing processes at room temperature and normal pressure whenever possible.
Use of Renewable Feedstocks:
Choose materials that come from renewable sources instead of ones that could run out.
Reduce Derivatives:
Use less energy by doing processes at room temperature and normal pressure whenever possible.
Catalysis:
Use catalysts, which are more efficient and create less waste, instead of other types of chemicals.
Design for Degradation:
Make chemicals that break down into harmless substances after use.
Real-Time Analysis for Pollution Prevention:
Check processes while they’re happening to catch and prevent harmful substances from forming.
Inherently Safer Chemistry for Accident Prevention:
Design processes to lower the risk of accidents like fires or spills.
Use of photochemistry and electrochemistry to drive green chemistry
The use of photons and electrons in green chemistry enables sustainable and efficient chemical reactions by eliminating the need for harmful reagents or excess energy. Photons, harnessed in photochemical reactions, activate molecules using light, often under mild conditions and without toxic chemicals. Similarly, electrons are utilized in electrochemical processes to drive reactions directly through electricity, avoiding the use of hazardous reducing or oxidizing agents. These methods not only minimize waste but also rely on renewable energy sources, such as sunlight or clean electricity, making them valuable tools for achieving greener, safer, and more energy-efficient chemical transformations.
Examples of sustainable Practices
Chemists use these principles in many ways to make their work safer and more sustainable. For example, they:
- Design products that aren’t harmful to people or the environment.
- Use renewable materials to make chemicals.
- Choose safer solvents or avoid them altogether.
- Develop processes that use less energy.
- Check their work in real-time to catch pollution early.
- Plan processes that are safer and less likely to cause accidents.
By preventing problems before they happen, green chemistry saves time and resources. This approach not only helps the environment but also makes chemical production more efficient.
Technology enabling Green Chemistry
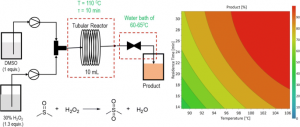
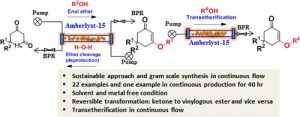
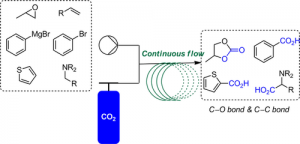
Technology, like Flow chemistry, makes green chemistry even easier to follow. Flow processes are safer because they use smaller amounts of chemicals and reduce waste. They also lower the chances of harmful chemicals escaping into the environment. Plus, flow processes can be scaled up easily without needing to start over. As a result, flow chemistry supports many of the 12 principles of green chemistry.