What is peptide synthesis?
Peptide synthesis is a process that produces peptides. Peptides are organic molecules consisting of between 2 and 50 amino acids linked by peptide (amide) bonds. Many natural processes produce peptides. Chemists have borrowed these natural processes in bacteria to produce the peptide that they need. However, the scope of this approach is limited. More recently, peptide chemists have used solid-phase peptide synthesis (SPPS) to produce difficult and unnatural sequences of peptides synthetically in a more controlled manner. Instruments have been designed to automate the process of solid-phase peptide synthesis. These instruments are called peptide synthesizers.
History of solid phase peptide synthesis (SPPS)
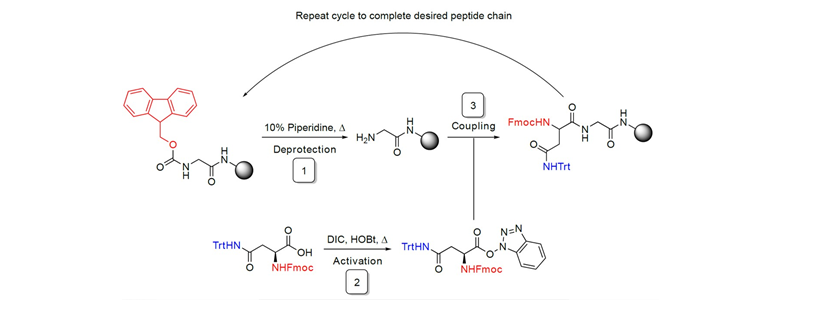
In 1963 a ground-breaking publication by Merrifield revolutionised peptide synthesis. Merrifield and his team began the synthesis of a peptide sequence by attaching the first amino acid to a solid polymer support.1 The rest of the sequence was built by deprotecting the terminal amine and adding the next protected amino acid in the sequence. Because the growing peptide is tethered to the solid support at one end, it is simple to wash away the excess of any unreacted monomer. The next deprotection and coupling stage begins with the newly cleaned peptide chain. Merrifield’s solid-phase approach set the stage for the production of complex and unnatural peptide sequences. Over the past 50 years, the process has been significantly optimised.
Peptide synthesis today
Today, the focus of therapeutic pharmaceuticals is beginning to shift away from small molecular medicines and towards larger, peptide-based treatments. Peptides have enormous potential as medicinal compounds. They are highly specific and often potent. Pharmaceutical groups have been attracted by the opportunity to modify peptide sequences to introduce greater physiochemical stability and solubility. As of 2016 there are over 50 peptide drugs in the market (non-insulin derived) with approximately 170 in various stages of clinical development.2 Total sales of peptide drugs in 2015 are estimated to be approximately $50 billion.3 Currently, most commercial peptide synthesis is performed in sequential or multi-parallel peptide synthesizers with a scale ranging from 1 µmol to 30 mmol. This peptide synthesis is often followed by a chromatographic purification. Bespoke batch vessels are often used for a clinically viable therapeutic peptide.
Benefits of continuous flow chemistry for peptide synthesis
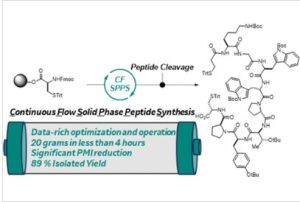
With over 20 years of expertise in flow chemistry Vapourtec is changing the peptide synthesizer market. Already adopted by several pharmaceutical and biotechnology companies, as well as academic institutes, Vapourtec’s peptide synthesis product portfolio is based on Fast Flow Solid Phase Peptide Synthesis (FF-SPPS). So why should anyone move their peptide synthesis to FF-SPPS?
Eliminating randomness, increasing crude purity
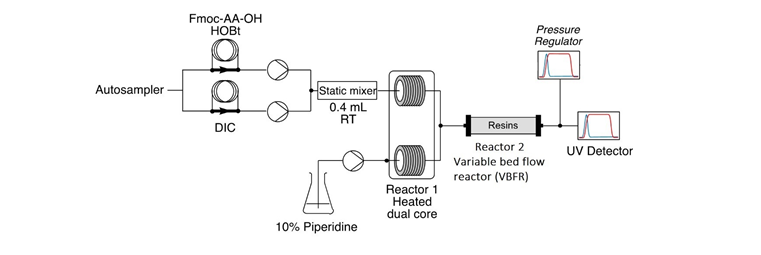
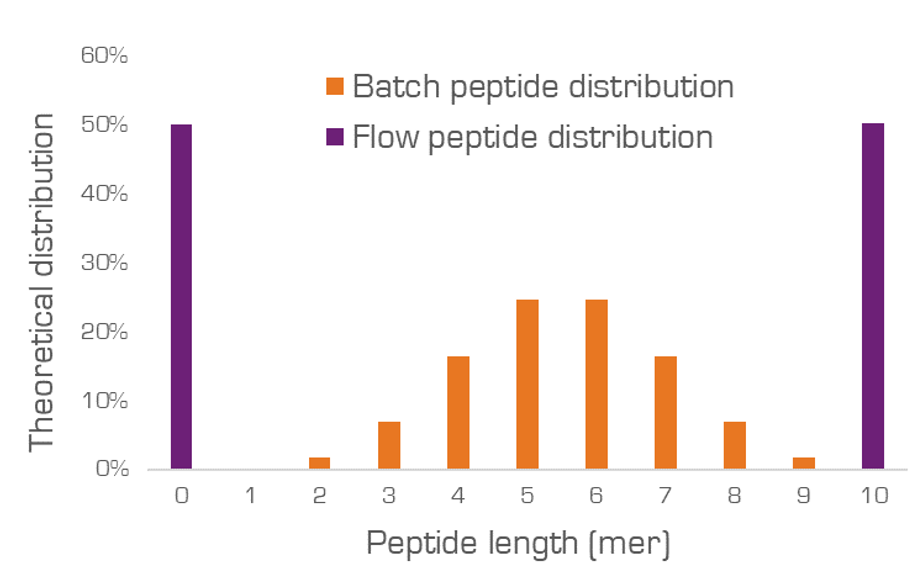
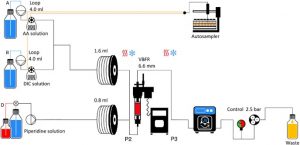
One of the key characteristics that differentiates FF-SPPS from a batch synthesis or recirculating flow synthesis, is the arrangement of the solid media. In batch, solid media is stirred in a vessel, continuously mixing. Reagents are then added to the vessel for the reaction to occur. The coupling process is random, and with every coupling the impurity profile increases to give a normal distribution of deletions (Figure 1).
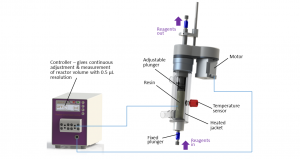
In continuous flow, the solid media is packed in Vapourtec’s proprietary Variable Bed Flow Reactor (VBFR) while reagents single pass the reactor. The VBFR is a perfect packed bed flow reactor that will automatically vary in volume to accommodate changes in the volume of the resin (increasing as amino acids are coupled, decreasing as Fmoc deprotection occurs). This inhibits the movement of resin beads while minimizing the reactor volume throughout the whole synthesis. This ensures a unique interaction between the reagents and the static resin, back mixing is eliminated, and reaction by-products are continuously removed. High reaction efficiency is achieved, but more importantly, by constraining both the resin and the direction of the reagent flow, the target peptide is preferred even at substoichiometric conditions.
Figure 1. Theoretical peptide distribution in batch vs flow for a 10-mer target peptide when all amino acids are used at 0.5 equivalents relative to the total resin loading.
Heated reactions to minimize aggregation and access difficult peptides
Heating has two important benefits: preventing β-sheet structures that can lead to aggregation events and increasing reaction kinetics which can be particularly beneficial for sterically difficult couplings. Using flow chemistry, it is straightforward to pre-heat and pre-activate the amino acids before they contact the resin. This ensures fast, more efficient coupling cycles as only activated amino acids will flow through the VBFR. However, not all heating is the same. Our experience supporting continuous flow applications over the last 20 years has taught us that uniform and constant heating is the key to reproducibility. Hot spots or uncontrolled temperature spikes cause racemization and can lead to loss of the ligand, particularly with chlorotrityl type resins.
Effortless scale-up with identical crude purity
Current batch technologies can require weeks of optimization and scale up of a peptide synthesis to produce large quantities for clinical studies.
Vapourtec developed the PS-30™ pilot scale peptide synthesizer to significantly reduce the time to market for development of new peptide-based therapeutics. This is achieved by using large scale reactors with identical heating and mixing characteristics as the laboratory scale platforms. Scientists can now optimize a synthesis at 50 µmol scale, and bring the same reaction conditions (equivalents, temperature and residence time) to a 30 mmol scale without further optimization.
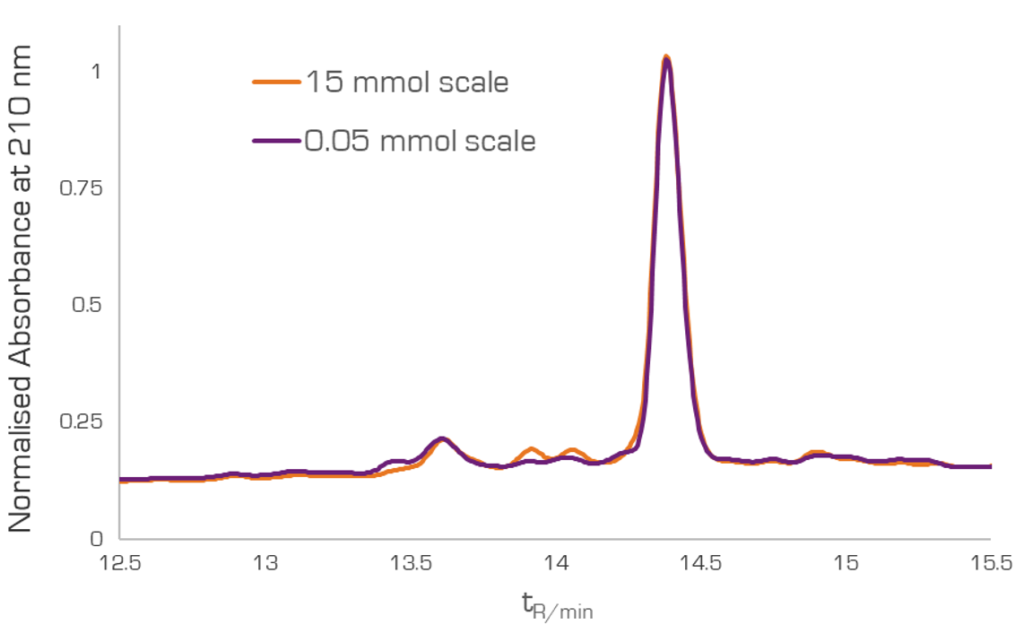
As an example, we used the PS-30 to scale up the synthesis of GLP-1 to 15 mmol in less than a day. The remarkable advantage is not only the synthesis time at this scale but also the identical crude purities achieved at both scales (Figure 2).
Figure 2. HPLC traces for the product of the GLP-1 synthesis at 50 µmol scale vs 15 mmol scale.
In-line data for easy reaction optimization
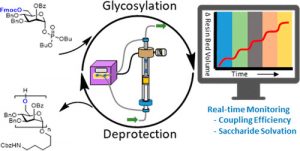
In addition to the chemical advantages, FF-SPPS provides real-time in-line data never seen before at that level of detail. Thanks to the VBFR, changes in the reactor volume can be recorded, which can help detect aggregation events, with precise timing. Also, rather than using UV spectroscopy to monitor a reaction sample or the waste stream as it would be done in batch, flow SPPS allows for quantitative real-time in-line analysis of Fmoc deprotection. Both sets of data can help with the immediate identification of any non-standard coupling events, allowing the user to recognize and optimize difficult coupling events at a small scale, before scaling up.
Further benefits
The VBFR technology together with the uniform heating allow for effective couplings with as low as 1.2 equivalents of amino acids. This translates into reduced costs, especially when using unnatural amino acids. FF-SPPS also benefits from an inherent advantage of flow chemistry, reducing solvent usage compared to batch techniques with only 60 ml/mmol per cycle.
The Vapourtec peptide synthesizer range
Vapourtec offer a range of fully automated FF-SPPS peptide synthesizers covering scales between 50 µmol and 30 mmol.
At the lab scale, the Peptide-Builder™ increases throughput within a small footprint with up to 16 automated peptides synthesized sequentially, the first peptide available within 2 hours (for a 16-mer). At the pilot scale, the PS-30™ pilot scale peptide synthesizer can potentially speed up the development of peptide drugs, with linear scale-up and synthesis times under a day.
In 2019 Vapourtec formed a collaboration towards peptide synthesis with Professor Peter Seeberger and his Group at Max Plank Institute, Berlin. The purpose of this collaboration was to synthesis a range of peptides with sequences known to have challenges caused by aggregation and resulting loss of solubility, obtaining data from the variable bed flow reactor (VBFR) and using the data obtained to optimise the synthesis conditions to improve the crude purity. This work has been published in the paper shown immediately below.
At Vapourtec, during a collaboration with New Path Molecular, we have optimised and scaled-up the synthesis of ACP (65-74). We found that the optimised conditions translated to the large-scale synthesis without any effect on the purity of the crude peptide (94%). This approach makes it possible for the user to use a small amount of resin for an initial synthesis and optimisation. The optimised conditions can be translated into the now well understood large scale synthesis.
Flow chemistry SPPS white paper
References
[1] Merrifield, R. B., J. Am. Chem. Soc., 1963, 85(14), 2149-2154
[2] Henniot, A., Collins, J. C., Nuss, J. M., J. Med. Chem., 2017, DOI: 10.1021/acs.jmedchem.7b00318
[3] Peptide Therapeutics Market: Global Industry Analysis, Size, Share, Growth, Trends and Forecast 2012-2018, Transparency Market Research,
https://www.transparencymarketresearch.com/peptide-therapeutics-market.html,[Accessed Nov. 2017]