Total Synthesis of Phytotoxic Radulanin A Facilitated by the Photochemical Ring Expansion of a 2,2-Dimethylchromene in Flow
- Bruce Lockett-Waltersa
- Simon Thuilliera,b
- Emmanuel Baudouinb
- Bastien Nay*a
- aLaboratoire de Synthèse Organique, Ecole Polytechnique, CNRS, Institut Polytechnique de Paris, 91128 Palaiseau, France
- bInstitut de Biologie Paris-Seine (IBPS), Laboratoire de Biologie du Développement, Sorbonne Université, CNRS, UMR7622, F-75005 Paris, France
 Read the publication that featured this abstract
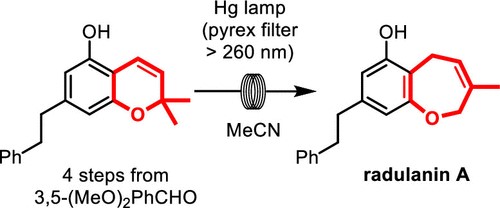
Read the publication that featured this abstractThe radulanins are biologically active bibenzyl natural products featuring a synthetically challenging 2,5-dihydro-1-benzoxepine core. In contrast with previous reports exhibiting lengthy strategies, we demonstrate the shortest synthesis of radulanin A to date, featuring a largely unexplored photochemical ring expansion reaction of a 2,2-dimethylchromene precursor. This work was adapted to a continuous-flow setup for larger-scale preparation, in view of biological investigations into the herbicidal properties of this natural product.
Get in touch
For more information on flow chemistry systems and services please use the contact methods below.
Call us on +44 (0)1284 728659 or Email us
Resource Centre
R-Series

The Vapourtec R-Series is, quite simply, unrivalled for flow chemistry
- Flexible |
- Precise |
- Automatable
The R-Series is undoubtedly the most versatile, modular flow chemistry system available today.
E-Series

The Vapourtec E-Series is the perfect introductory system for flow chemistry
- Robust |
- Easy to use |
- Affordable
The E-Series is a robust and affordable, entry level flow chemistry system designed for reliability and ease of use.
