Date: 26 September 2022 | Category: News
In the recent paper entitled “Multicomponent Direct Assembly of N-Heterospirocycles Facilitated by Visible-Light-Driven Photocatalysis” (1), the Ley group demonstrates the use of the Vapourtec UV-150 photochemical reactor in the synthesis of complex spirocyclic compounds under mild conditions.
The interest of spirocyclic scaffolds in medicinal chemistry has risen in the past decades, but the main obstacle has been the challenging synthetic steps required to yield those compounds.
Existing methods typically require several steps and the use of expensive catalysts, like Ruthenium or Rhodium, to obtain the desired product.
One of the solutions to simplify the synthetic route was to adopt reagentless chemistry approaches. Photochemically, the generation of nitrogen-centered radicals is relatively simple and proved to activate N-H and N-X bonds. By utilising this methodology in the synthesis of spirocyclic compounds, steric issues caused by quaternary carbon centres can be avoided, improving the overall process.
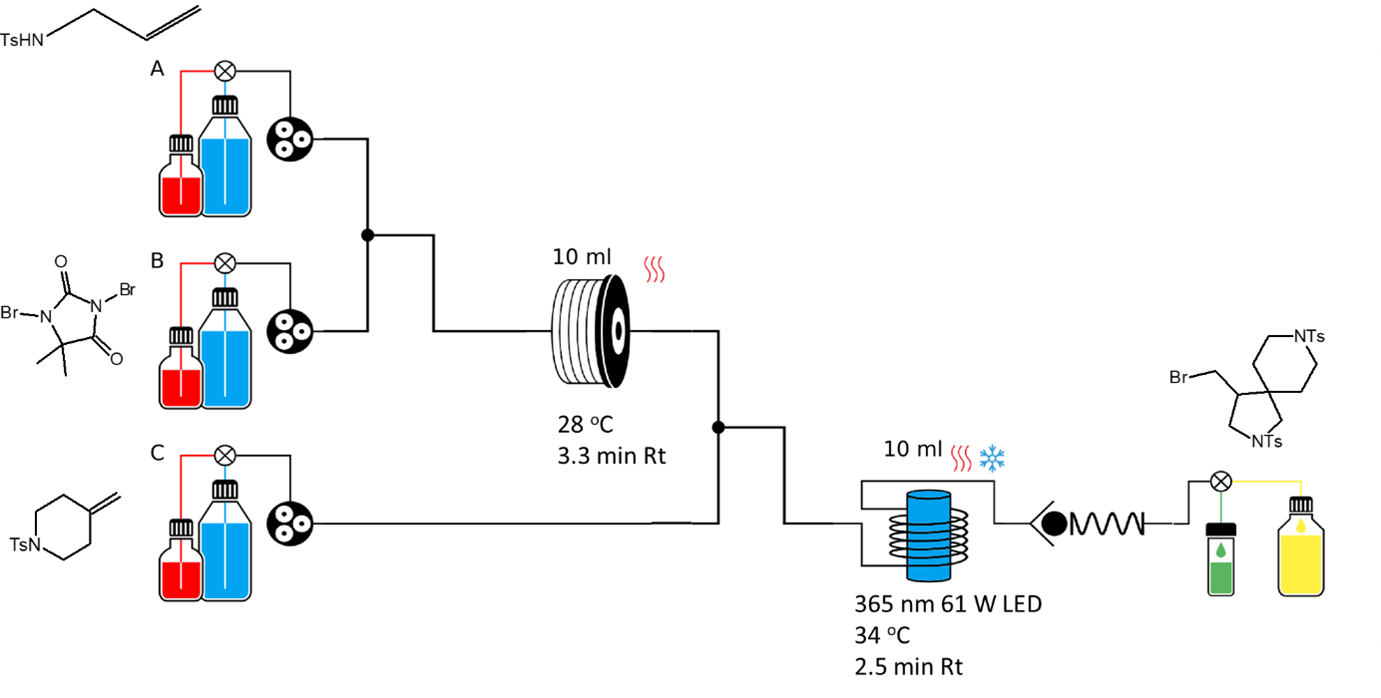
Using an easy-Photochem equipped with a 365 nm high power LED light, Mr. Griffiths and Prof. Ley illustrate the synthesis of a wide array of spirocyclic pyrrolidines. Remarkably for some substrates, the protocol worked with inexpensive organic photocatalysts.
This methodology was scaled up using the Photochemical reactor UV-150 in the synthesis of the bis-azaheterospirocycle detailed below, achieving a throughput of 100 g/day.
1) O.M.Griffiths and S.V.Ley, J.Org.Chem, 2022 published on line at https://pubs.acs.org/doi/10.1021/acs.joc.2c01684
To read the publication featured click here
To find out more about the UV-150 photochemical reactor click here
