Date: 2 April 2026 | Category: News
Authors: Andrew J. S. Lin, Nicholas S. O’Brien, Abigail Florence, Matthew Killen, Shelby L. Frailey, Jayne Gilbert, Jennette A. Sakoff, Mohammed K. Amin, Emily E. Castelloe, Ngoc Chau, Jing Xue, Phillip J. Robinson, and Adam McCluskey*
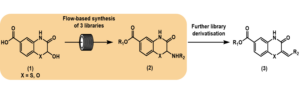
The McClusky group at the University of Newcastle in New South Wales, Australia, has used the Vapourtec E-Series easy-MedChem system in the synthesis of a library of substituted benzylidene-3-oxo-3,4-dihydro-2H-benzo[b][1,4]thiazine-6-carboxylic acid (1) derivatives.[1]
Dynamin GTPase and its role in endocytosis
Cells have developed key processes where various external materials, such as macromolecules and small particles, can be absorbed from the cell surface into a cell. This process – endocytosis – is crucial for cell function. One of the main endocytosis pathways is clathrin-mediated endocytosis, or CME. In this process, clathrin-coated vesicles are formed on the surface of the cell and then pass through the cell membrane into the cell. At this point, a GTPase, in this case dynamin, severs the vesicle from the plasma membrane and the contents of the vesicle are released and taken to the appropriate cellular target. CME is a key process in vesicular trafficking, but defects in this pathway have been implicated in a range of diseases including Alzheimer’s, epilepsy, and drug resistance in cancer. Furthermore, inhibition of the CME pathway is touted as a possible therapeutic target for treatment of chronic pain, epilepsy, cancer, and viral infection.
Dynamin is a large GTPase of around 100 kDa, comprising three gene families, DNM1, DNM2, and DNM3, which code for dyn I, dyn II, and dyn III, respectively. Inhibition of dynamin, for example through use of prochlorperazine (Stemetil) and chlorpromazine, has been shown to restore antibody effectiveness in head and neck cancer trials.[2]
Continuous flow chemistry and rapid library synthesis
During this work, a continuous flow approach was used to rapidly produce materials that served as the basis for three distinct compound libraries, and a further three compound libraries were produced using batch methods. Screening of the libraries showed that the position of the carboxylate moiety at C6 was essential for activity, and H-bond donor/acceptor capability on the left-hand aromatic ring was also beneficial in cell free assays. Several new inhibitors of dynamin were disclosed, with ≤10 μM inhibition, Figure 1.
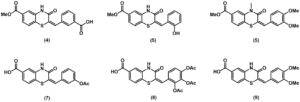 Figure 1: A selection of the new inhibitors of dynamin discovered in this work with ≤10 μM inhibition
Figure 1: A selection of the new inhibitors of dynamin discovered in this work with ≤10 μM inhibition
Summary
Overall, a combination of continuous flow and batch chemistry were used to build a series of libraries for inhibition of dynamin, a key protein in CME and downstream cell processes. Use of the Vapourtec E-Series easy-MedChem system facilitated this work, giving rapid access to key intermediates.
References:
[1] Substituted Benzylidene-3-Oxo-3,4-Dihydro-2H-Benzo[b][1,4]thiazine-6-Carboxylic Acid Analogs as Dynamin GTPase Inhibitors (A. J. S. Lin, N. S. O’Brien, A. Florence, M. Killen, S. L. Frailey, J. Gilbert, J. A. Sakoff, M. K. Amin, E. E. Castelloe, N. Chau, J. Xue, P. J. Robinson, A. McCluskey, Chem. Med. Chem., 2026, 21, e202500377). https://doi.org/10.1002/cmdc.202500377
[2] Endocytosis Inhibition in Humans to Improve Responses to ADCC-Mediating Antibodies (H. Y. Chew; P. O. D. Lima; J. L. G. Cruz; B. Banushi; et al., Cell 2020, 180, 895). https://doi.org/10.1016/j.cell.2020.02.019