Date: 29 August 2025 | Category: News
The rise of GLP-1 receptor agonists (GLP-1 RAs) has been meteoric. Initially targeted towards Type 2 diabetes, the significant weight loss benefits observed with GLP-1 RAs such as semaglutide (Ozempic®, Wegovy®), liraglutide (Victoza®, Saxenda ®) and tirzepatide (Mounjaro®) have led to these treatments becoming blockbuster weight management therapies as well as effective diabetes medications. This sudden surge in demand has outpaced the production capacity of these peptides for several years. For example, semaglutide has suffered from limited supply in the US since 2022 due to its unprecedented demand, with these issues only recently being resolved [1].
What are GLP-1 agonists?
When food is ingested, the human body releases a hormone called glucagon-like peptide-1 (GLP-1), which instigates the release of insulin in hyperglycemic states, reduces glucagon secretion in hyperglycemic or euglycemic (normal) states, and also slows stomach emptying, all of which modulate glucose homeostasis (2). As well as being a key player during digestion, GLP-1 is also known to modulate areas of the brain involved in appetite, leading to significant appetite suppression and subsequent effects on weight loss through reduction of food intake. GLP-1 is broken down within 1 to 2 minutes of release in the body by the body’s enzymatic system, because it breaks down so quickly, native GLP-1 is not therapeutically useful as a drug.
GLP-1 receptor agonists (GLP-1RAs) are chemical structures, largely based on peptides, that can mimic GLP-1 hormone through binding to key receptors in the body, producing the same effects. However, GLP-1RAs are usually modified analogues that are designed to be much longer acting, with a therapeutic window of up to 1 week, and their use is rapidly becoming a key treatment for patients with type 2 diabetes as well as those with a requirement for weight loss.
With the popularity of GLP-1 agonists ever-increasing and over 150 GLP-1 drug candidates in development [3], the field is continually expanding, most recently into oncology and obesity-related cancers.[4] While most approved GLP-1RAs require subcutaneous injection, substantial work is being undertaken to move toward oral GLP-1 therapies [5], such as Rybelsus which was the first oral GLP-1RA approved by the FDA in 2019. Research is also expanding beyond targeting GLP-1 alone; the obesity treatment retatrutide targets GLP-1 in combination with glucose-dependent insulinotropic polypeptide (GIP), and glucagon GCG receptors [6] to produce substantial weight loss.
In light of this increased interest, pharmaceutical research and development teams have been forced to adapt manufacturing processes to access these complex peptides more rapidly and cost-effectively than ever before.
Complex synthetic challenges – particularly at scale
The agonist molecules themselves are complex peptides, each containing over 30 amino acids. For example, semaglutide is composed of 31 amino acids (Figure 1), and tirzepatide is even larger with 39 amino acid residues.
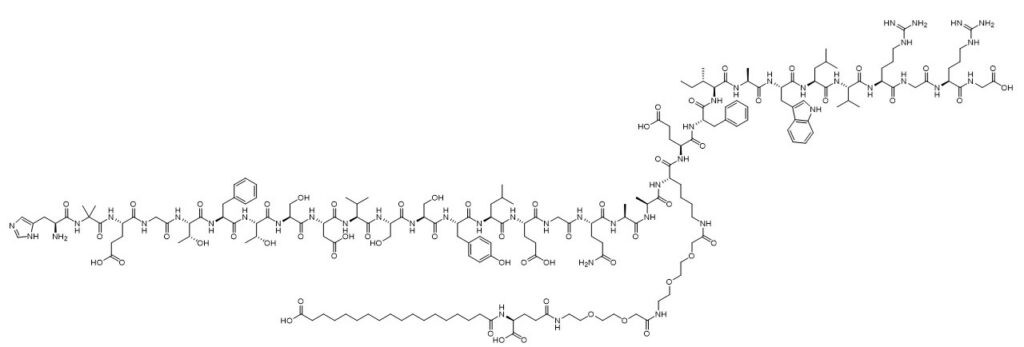
Figure 1: Chemical structure of semaglutide
Building these peptide sequences requires specialist knowledge and equipment. Current techniques include using sequential or multi-parallel peptide synthesizers that exploit solid phase peptide synthesis (SPPS). While useful and still heavily relied upon, these traditional batch synthesis approaches can suffer from significant issues including complex impurity profiles due to incomplete reactions or double-addition, and significant challenges accessing “difficult sequences” [7], such as highly hydrophobic peptides that are prone to aggregation.
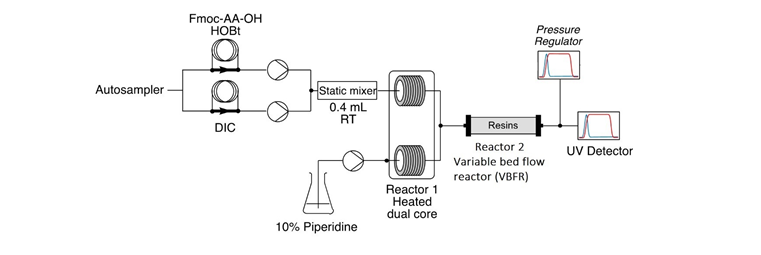
Vapourtec’s Continuous Flow Peptide Synthesizer: Solving synthesis bottlenecks
Vapourtec’s breakthrough family of peptide synthesizer products harnesses Fast- Flow Solid Phase Peptide Synthesis (FF-SPPS) technology [8] to produce polypeptides with improved purity and with significant time savings, Figure 2.
Using continuous flow, the solid media is packed in Vapourtec’s proprietary Variable Bed Flow Reactor (VBFR) and reagents are passed in sequence through the reactor. This achieves a unique interaction between the reagent and the resin, with reaction by-products continuously removed giving a high-efficiency process. The design of the product also facilitates exquisite control of temperature; reagents can be pre-heated or pre-activated before contact with the resin, with use of higher temperatures often minimizing aggregation and facilitating access to sterically challenging couplings.
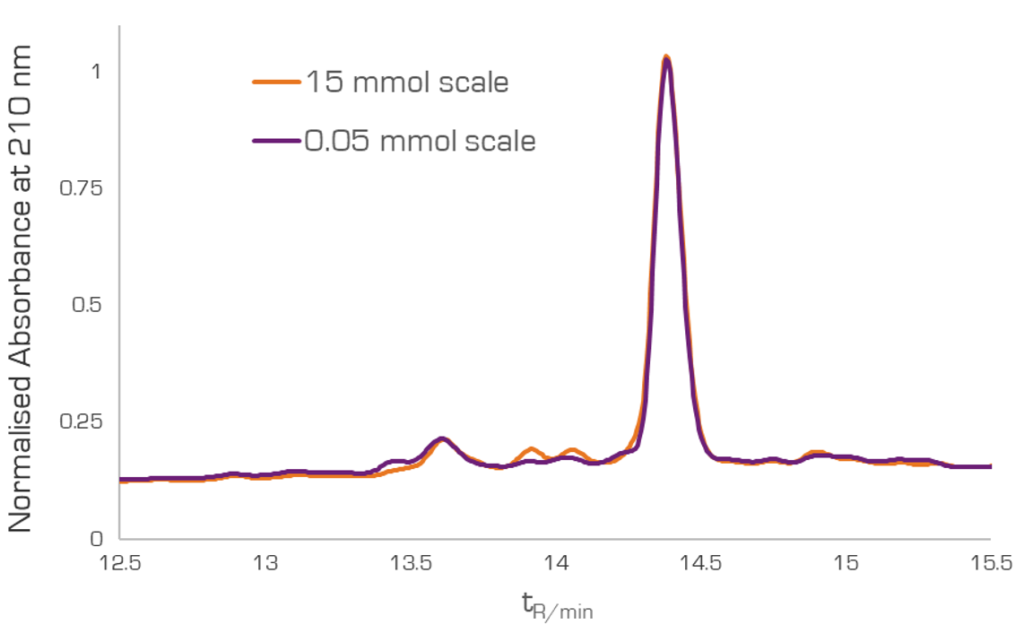
When a peptide synthesis requires scale-up, Vapourtec’s PS-30™ pilot scale peptide synthesizer uses large scale reactors that provide identical heating and mixing characteristics to the laboratory-scale platforms, enabling rapid access to up to 30 mmol scale without further optimization. For example, the synthesis of GLP-1-based 29-mer analogy was rapidly scaled from 0.05 mmol to 15 mmol with an identical crude purity using the PS-30TM, Figure 3. In addition, solvent use was minimized and reaction time only slightly increased (this 29-mer peptide was synthesised at 15 mmol scale in only 15 hours).
Vapourtec: Your partner for peptide innovation
As the field of peptide drug discovery gathers momentum and expands beyond GLP-1 [9], find out how Vapourtec’s proprietary peptide technologies can expedite your peptide discovery and deliver the next generation of breakthrough peptide therapeutics on scale and at speed. Explore our peptide synthesizer product family here.
References:
[1] FDA clarifies policies for compounders as national GLP-1 supply begins to stabilize (Accessed August 2025). https://www.fda.gov/drugs/drug-safety-and-availability/fda-clarifies-policies-compounders-national-glp-1-supply-begins-stabilize
[2] Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes (J. P. Frías, M. J. Davies, J. Rosenstock et al., N. Engl. J. Med., 2021, 385, 505–515). https://doi.org/10.1056/NEJMoa2107519
[3] GLP-1 Market Industry Trends and Global Forecasts 2024-2035: Over 150 GLP-1 Drug Candidates in Development for Multiple Indications, Type 2 Diabetes and Obesity Drive GLP-1 Drug Development Boom (Accessed July August 2025). https://www.globenewswire.com/news-release/2025/03/18/3044263/0/en/GLP-1-Market-Industry-Trends-and-Global-Forecasts-2024-2035-Over-150-GLP-1-Drug-Candidates-in-Development-for-Multiple-Indications-Type-2-Diabetes-and-Obesity-Drive-GLP-1-Drug-Deve.html
[4] GLP-1 Drugs: A Promising New Frontier for Cancer Risk Reduction (Accessed July August 2025). https://www.drugdiscoverynews.com/glp-1-drugs-mimic-the-anti-cancer-benefits-of-bariatric-metabolic-surgery-16497
[5] Experimental GLP-1 pill helped people with obesity lose weight, Eli Lilly says (Accessed August 2025). https://www.cnn.com/2025/08/07/health/lilly-glp1-pill-weight-loss
[6] Triple–Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial (A. M. Jastreboff, L. M. Kaplan, J. P Frías et al., N. Engl. J. Med., 2023, 389, 514–526). https://doi.org/ 10.1056/NEJMoa2301972
[7] Challenges and Perspectives in Chemical Synthesis of Highly Hydrophobic Peptides (L. K. Mueller, A. C. Baumruck, H. Zhdanova, A. A. Tietze, Front. Bioeng. Biotechnol., 2020, 8). https://doi.org/10.3389/fbioe.2020.00162
[8] Solid phase peptide synthesis (Accessed August 2025). https://www.vapourtec.com/applications-of-flow-chemistry/solid-phase-peptide-synthesis/
[9] The Current State of Peptide Drug Discovery: Back to the Future? (A. Henninot, J. C. Collins, J. M. Nuss, J. Med. Chem., 2018, 61, 1382–1414). https://doi.org/10.1021/acs.jmedchem.7b00318