Date: 4 June 2014 | Category: News
Using the Vapourtec R-Series flow chemistry system researchers from Janssen Research and Development (Spain), the University of Leuven and the Universidad de Castilla-La Macncha have shown a continuous flow process for carbonylation reactions without the need of toxic carbon monoxide gas and have shown new chemical reactivity under flow conditions that were not previously possible under batch conditions.
Continuous flow chemistry has developed to be a useful enabling technology that allows access to novel process windows giving excellent control of reaction parameters. The use of flow chemistry techniques allow safer handling of hazardous reactants through the containment and reaction of smaller inventories of material and in some cases the reduction of these dangerous chemicals while allowing the reaction to be easily scaled.
Palladium catalysised carbonylation reactions using carbon monoxide is a key reaction in organic synthesis and in the bulk chemical industry. The use of CO, despite its applicability is a serious safety issue due to its toxic nature and reduces the utility of these reactions in research laboratories that don’t have the associated safe working environments. This is the first example of performing carbon monoxide coupling reactions under flow conditions using a precursor molecules that releases CO as a mild, safe and desirable alternative to CO gas.
The researchers describe the use of aryl formates as a CO precursor with a range of (pseudo) haloarenes for the first time. Under these mild conditions reaction times were reduced from hours to minutes, catalyst loading was reduced and the amount of toxic CO reduced to near equimolar amounts.
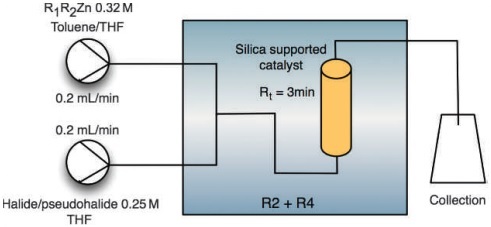
Under these conditions the authors were able to demonstrate the selective reaction of 3,4-dibromothiphene to the monoester where only the diester could be obtained in batch experiments.
Find out more Published papersView the full Journal of Flow Chemistry paper.
