
Date: 17 September 2014 | Category: News
Using the Vapourtec R-Series flow chemistry system researchers from the Max-Planck Institute of Colloids and Interfaces have developed an inexpensive, large scale synthesis of a range of anti-Malaria drugs under continuous flow conditions. The continuous, divergent synthesis system is coupled to continuous purification and is capable of producing four anti-malarial APIs. The system is comprised of three linked reaction modules for photo oxidation/cyclization, reduction, and derivatization. A fourth module couples the crude reaction stream with continuous purification to yield pure API. The output from the purification step is pure enough to meet WHO and FDA guidelines.
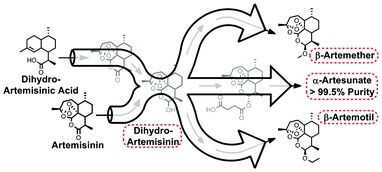
The method which has been developed allows for the complete production of anti-malaria medicines in a continuous process in one location. The process uses both artemisinin and the plant waste product generated to produce these medicines without having to increase the amount of material farmed. This ability to use multiple sources for production expands on the group’s previous work showing the synthesis of artemisinin from artemisinic acid under photochemical conditions.
“The new method will allow for the possibility to transfer further steps of the value chain to developing countries, which currently only grow and extract the plant,” says Dr. Kerry Gilmore, group leader of the Continuous Chemical Systems group in Prof. Seeberger’s team. “More importantly, this can help to dramatically shorten the supply chain and increase the capabilities of developing countries with respect to their pharmaceutical independence.”
Peter H. Seeberger: “We think our approach could be the best solution to lower the cost of anti-malarial production. We can use all major substances in the plant, our method is cheaper than all others, and it produces very pure medications. On the one hand we can transform the artemisinin produced by extractors into the medications, while on the other hand we can produce medications from the waste of that same extraction. Comparatively we produce about two times as much medication from the plant as previously possible and we help the farmers and extractors in the developing countries to make a living from the plant.”
This continuous modular approach has a number of advantages over the equivalent batch process which are more labour intensive, require purification and solvent removal after each step and increase the exposure to the hazardous reagents used in the synthesis.
Find out more Published papersRead the article on the the Chemistry World website
Read the Chemical Communications paper
