Date: 12 March 2026 | Category: News
Synthetic Pathway to trans-A2B2‑Porphyrins: From Oxime-Substituted Dipyrromethanes to Functionalized Macrocycles
Authors: João C. S. Simões, Bruna D. P. Costa, Ana Clara B. Rodrigues, Susana M. M. Lopes, J. Sérgio Seixas de Melo, Marta Pineiro, and Teresa M. V. D. Pinho e Melo
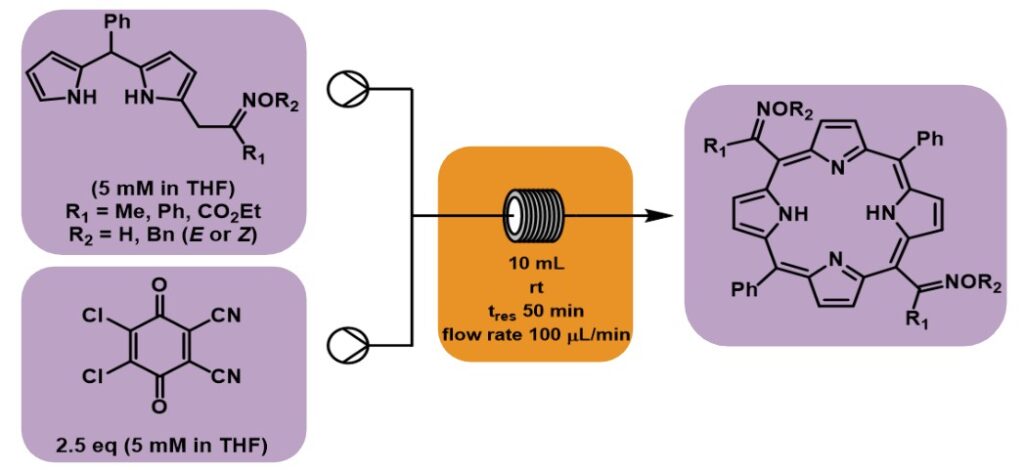
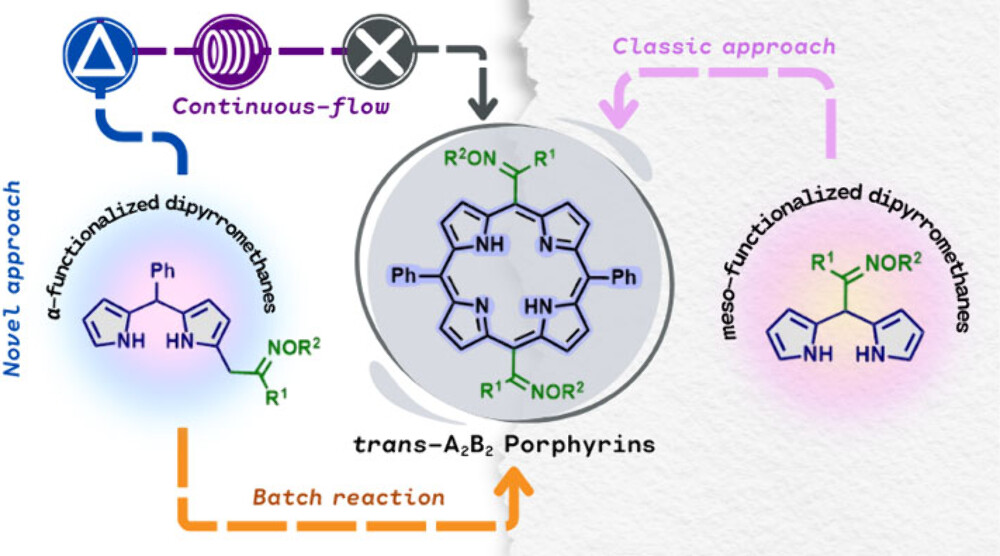
Researchers from the group led by Teresa M. V. D. Pinho e Melo at University of Coimbra, Portugal, optimised in batch and flow – for which they used the Vapourtec E-Series easy-Medchem system – the preparation of trans-A2B2-porphyrins from α-oxime-functionalized dipyrromethanes.[1] In particular, the continuous flow process was considered more sustainable, and led to fewer side products.
Porphyrins synthesis: tuning structure impacts photophysical properties
The porphyrins synthesised in this work were not the originally intended products, however their formation was noted as a minor product in initial synthetic investigations towards BODIPY derivatives. Investigation into porphyrin formation and photophysical properties revealed the opportunity to develop a route for porphyrin synthesis orthogonal to the typical condensation of pyrrole derivatives with aldehydes or ketones in the presence of catalysts. It was found that the inclusion of DDQ was essential for the oxidative coupling of two α-oxime-difunctionalized dipyrromethanes, with a radical pathway postulated as the reaction mechanism.
While the initial reaction conditions were developed using batch chemistry, a more scalable and sustainable approach was developed using flow chemistry. Overall, a simple flow chemistry synthesis of functionalized Porphyrins using a Vapourtec system led to optimised synthesis and purification of materials, with promising applications in materials science and medicine.
References:
[1] Synthetic Pathway to trans-A2B2‑Porphyrins: From Oxime-Substituted Dipyrromethanes to Functionalized Macrocycles (J. C. S. Simões, B. D. P. Costa, A. C. B. Rodrigues, S. M. M. Lopes, J. S. Seixas de Melo, M. Pineiro, T. M. V. D. Pinho e Melo*, J. Org. Chem., 2025, 90, 16060–16069). https://doi.org/10.1021/acs.joc.5c01907