Date: 26 February 2026 | Category: News
Sagar Arepally, Hanaa Gieman, and Thomas Wirth
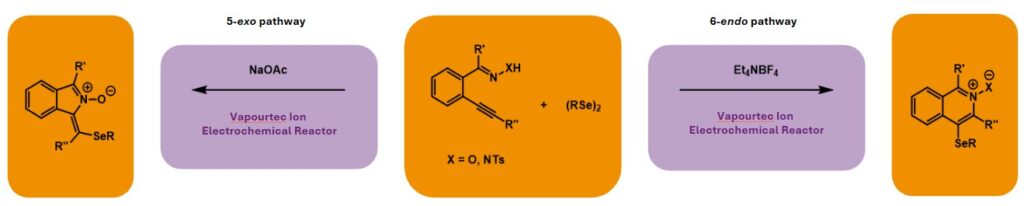
The Wirth group at Cardiff University have successfully used the Vapourtec Ion electrochemical reactor to effect electroselenocyclisations under continuous flow conditions.[1] Key to reaction success was the identity of the electrolyte, which, if changed, could be used to tune the electrocyclisation to furnish isoindole N-oxides (via a 5-exo pathway) or isoquinoline 2-oxides (via a 6-endo pathway). Use of flow chemistry allowed for facile scale-up, with gram-quantities of N-activated isoquinoline derivatives prepared, which were subsequently functionalised to generate pharmaceutically relevant compounds.
Electrochemistry within organic synthesis
In recent years, electrochemistry has re-emerged as a useful tool within the toolbox of organic synthesis. [2] In particular, electrochemistry allows for the precise control over redox processes and generation of reactive intermediates through the use of an electric current or electrode potential, providing access to a diverse array of chemical transformations. [3] Alongside this, electrochemistry is considered to be sustainable due to the traceless nature of the electrons used to effect a chemical transformation. However, despite these advantages, the adoption of electrochemistry has not been widespread. This is likely because most experiments are still conducted in batch, where there are severe limitations including low productivity, high electrolyte concentrations, and difficulties in scaling up.
Flow chemistry provides a valuable alternative to batch chemistry for a variety of reasons, including temperature control, rapid optimisation, facile scale-up and control of any potential safety issues. The use of flow chemistry, particularly for photo- and electrochemical reactions, has provided significant advantage due to the exquisite control of reaction conditions and practical advantages, such as excellent mass transfer, shorter residence times and, in the case of electrochemistry, higher electrode surface-to-volume ratio. This means that reproducibility of reaction conditions is often higher, and side-reactions can be minimized.
Electrochemistry and the importance of the electrolyte
Traditionally, supporting electrolytes were solely regarded as ionic conductors, but in recent years this has started to change and they now have utility as both reagents and mediators. For example, these electrolytes have been used in substrate oxidations, predominantly when this step occurs first, or with halide anions present, to achieve specific reactivity. However, their use as a base to assist substrate oxidations has been underexplored. The Wirth group identified an opportunity to utilise electrolytes as bases in regiodivergent electrochemical synthesis, leading to the development of electroselenocyclisations of double and triple bonds to generate medicinally relevant isoindole derivatives.[1]
During optimisation, the use of flow chemistry facilitated rapid screening of conditions, for example through varying the electrolyte, charge, solvent, cathode, flow rate and subsequent residence time. Within the systems studied, the electrolyte had a notable impact upon the cyclisation pathway followed: NaOAc led to exclusive formation of isoindole N-oxide (5-exo), whereas Et4NBF4 favoured the isoquinoline 2-oxide (6-endo).
Formation of the isoquinoline 2-oxide had excellent functional group compatibility, with electron-donating and electron-withdrawing substituents on the alkyne tolerated, as well as TMS-protected alkynes. Substituents on the oxime were also tolerated, including challenging ortho-alkynylaryl aldoximes, which are known to oxidise under electrochemical conditions. Moreover, these aldoxime substrates provide a handle for further functionalisation in the C1 position. Installation of a tellurium atom, by use of diphenyl ditelluride, was also achieved. Finally, ortho-alkynylaryl hydrazides also underwent 6-endo cyclisation yielding isoquinolinium imides, albeit in lower yields.
Isoindole N-oxide formation was also general, with a range of aryl-substituted compounds converted smoothly and in good yield, including electron-rich and electron-poor species. In some cases, the conditions were adjusted, for example by using a modified solvent system or different current. However, ortho-alkenylaryl aldoximes and tosylhydrazides were not fruitful under these conditions.
Flow chemistry as a tool to access new functionality
Overall, the use of flow chemistry, in particular the Ion electrochemical reactor, provided advantage over a batch approach by enabling control of key reaction parameters and screening of conditions, as well as facilitating scale-up to a 3.0 and 5.0 mmol scale.
References:
[1] Electrolyte-Controlled Regiodivergent Continuous Flow Electroselenocyclisations (S. Arepally, H. Gieman, T. Wirth, Angew. Chem. Int. Ed., 2025, 64, e202509811). https://doi.org/10.1002/anie.202509811
[2] Powering the Future: How Can Electrochemistry Make a Difference in Organic Synthesis? (T. H. Meyer, I. Choi, C. Tian, L. Ackerman, Chem, 2020, 6, 2484–2496). https://doi.org/10.1016/j.chempr.2020.08.025
[3] Bridging Lab and Industry with Flow Electrochemistry (N. Tanbouza, T. Ollevier, K. Lam, iScience, 2020, 23, 101720). https://doi.org/10.1016/j.isci.2020.101720