Date: 17 April 2024 | Category: News
Vapourtec Chief Scientific Officer Dr. Manuel Nuño recently met with Prof. Thomas Wirth at the Flow Chemistry European Summit 2024 in Rotterdam, and it was the perfect occasion to ask him some questions about a recent high-profile publication in Science Advances entitled “Electric field–assisted anion-π catalysis on carbon nanotubes in electrochemical microfluidic devices” [1].
Prof. Wirth is one of the leading experts in continuous flow electrochemistry and his group has helped Vapourtec design the Ion electrochemical reactor, which is now very widely used for flow electrochemistry.
The research published in Science Advances by the Wirth group in collaboration with Prof. Stefan Matile’s group at the University of Geneva at the end of last year was an important breakthrough. Together they discovered how an external electric field can be used to control and accelerate a chemical reaction like a ‘‘switch” and did so using the Vapourtec Ion electrochemical reactor. We asked Prof. Wirth a few questions about this work so that every chemist – even those not experts in the field – can understand what was so outstanding about this achievement.
Dr Nuño: “How does this piece of research differ from traditional electrochemistry?”
Prof. Wirth: “Electrochemistry typically relies on the flow of electrons to promote reactions by either adding or removing electrons from the substrate. This new approach uses electricity to create an electric field to promote reactions. Unlike traditional electrochemistry, we used the Ion reactor to “switch” the reaction on and off. This electric field induces a macrodipole in the multiwalled carbon nanotubes (MWCNTs) which are attached to the electrode and that is what is driving the reaction.”
Dr Nuño: “And why does this have to be done in flow?”
Prof. Wirth: “Traditional electrochemistry cannot create such interactions since the distance between electrodes is too large, and additional electrolytes inhibit this macrodipole interaction. By working in flow, with the narrowest possible electrode gap, we managed to achieve electrolyte-free conditions allowing us to induce a macrodipole in MWCNTs, controlling and tuning the catalysed process for the first time using substantial quantities of the starting material.”
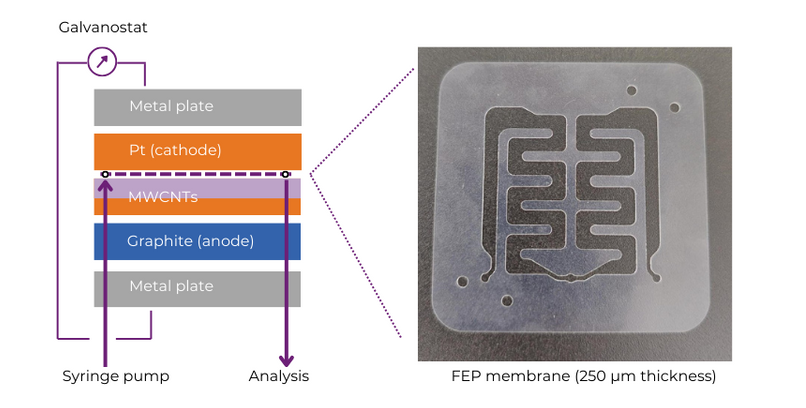
Experimental set-up using the Ion electrochemical reactor:
The Ion electrochemical reactor was assembled using a thin FEP membrane (250 µm thickness). This electrode gap is thin enough to allow the flow of electrons in electrolyte-free conditions. The electrode pair used was platinum and a modified graphite electrode, with MWCNTs drop cast on its surface.
Without any applied current, the reaction could not take place as there was no induced macrodipole to “switch on” the catalyst. With a low current, an oriented external electric field was generated, “switching on” the catalyst to promote epoxide-opening ether cyclizations. Although the investigated reaction is only serving to demonstrate the principle, new developments will include practical reactions towards interesting target molecules.
[1] Electric field–assisted anion-π catalysis on carbon nanotubes in electrochemical microfluidic devices
MA Gutiérrez López, R Ali, M-L Tan, N Sakai, T Wirth, S Matile, Sci. Adv. 9, eadj5502 (2023)
https://www.science.org/doi/10.1126/sciadv.adj5502
To find out more about the Ion electrochemical reactor, click here