Date: 30 April 2015 | Category: News
This application note demonstrates a green photochemical route to the reduction of Nitro compounds. The powerful combination of Vapourtec’s V-3 pump and the UV-150 Photochemical reactor is utilized to deliver the photo catalyst as a slurry and provide precise control over the continuous flow photochemical reaction, even with the short reaction times of less than 1 minute. The TiO2 photo catalyst is safe, cheap and simply removed by filtration. There is also a fascinating comparison of the performance of three different 365 nm light sources; filtered mercury lamp, LED Gen-1 and LED Gen-2. The Gen-2 LED achieves comparable throughput of the mercury lamp while consuming <50% of the power.
Although photochemistry is not a new science, its application in pharmaceutical and fine chemical manufacture has been limited, in part due to the lack of suitable equipment (especially at scale), but also due to poor efficiency for traditional batch reactors. The synergy of flow and photo chemistry offers greater benefit. Achieving uniform UV exposure in batch reactors is difficult, even if reactants are agitated. With flow chemistry, reactants are continuously passed by the UV source, removing product from the irradiated area, providing greater control and consistency, both in resultant compound and repeatability.
Recent developments in LEDs also offer huge potential for improving outcomes in photochemistry. High efficiency UV LED technology, particularly at longer wavelengths used in catalyst mediated reactions allows the source wavelengths to be tuned to the application. Reaction yield and selectivity is improved, as well as reducing waste heat and power consumption when compared to traditional mercury lamp technology. LED are safer in use, easier to maintain and unlike Mercury lamps can be used safely at scale without recourse to sophisticated inert gas blanketing systems.
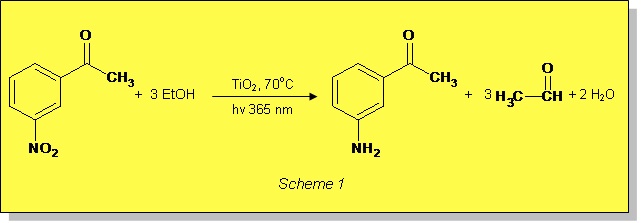
Conventional hydrogenations such as catalytic hydrogenation on metals or homogeneous catalysis using metal complexes are common reactions in organic chemistry. An alternative approach to hydrogenation is presented in the new application note using TiO2 as the photocatalyst, which offers a green alternative to produce the corresponding amines. The benefits include:
- The use of only ethanol as both the solvent and hydrogen source.
- The use of an easily removable, cheap and very safe catalyst, TiO2 which is commonly found in many commercial products, such as toothpaste, sunscreen and paints.
Reaction conditions used in photocatalytic TiO2 reductions only require mild conditions, making it an ideal transformation step in multi-step synthesis if the compound contains other reducible functions.
Download app note Find out more