Date: 12 September 2024 | Category: News
Lucas Kambanis, Anthony Ayoub, Max J. Bedding, Peter H. G. Egelund, Joshua W. C. Maxwell, Charlotte Franck, Lucien Lambrechts, Paige M. E. Hawkins, Timothy S. Chisholm, Joel P. Mackay, Emma Sierecki, Yann Gambin, Sameer S. Kulkarni, and Richard J. Payne*
The Payne group at the University of Sydney, Australia, have shown that a Vapourtec R-Series flow chemistry system can be used to effectively undertake expressed protein ligation (EPL), a closely related technique to native chemical ligation (NCL), which is used to enable the chemical synthesis of proteins. Preparation of tens of milligrams of high purity material significantly faster than in batch mode was possible, paving the way for the scale up of modified protein therapeutics through continuous flow processes.
Prof. Payne said, “The Vapourtec R-Series is perfectly suited to peptide ligation in flow and the ability to perform photochemistry in line has opened up a number of new opportunities for us in peptide and protein production.”
What is native chemical ligation (NCL)?
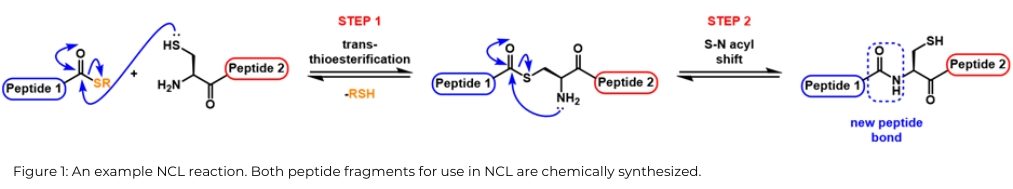
Since its disclosure by Kent and co-workers in 1994, native chemical ligation (NCL) has had substantial impact upon the chemical synthesis of proteins [1] and is extremely effective for synthesis of larger polypeptide chains or proteins, although generation of proteins with >200 amino acid residues is still a considerable challenge. NCL comprises two steps: transthioesterification, where the thiol side chain of cysteine nucleophilically attacks the carbonyl of a thioester to give a new thioester, and a rapid S–N acyl shift that forms the peptide bond and regenerates the native cysteine moiety, [Figure 1].
Expressed protein ligation (EPL) in flow as an alternative technology
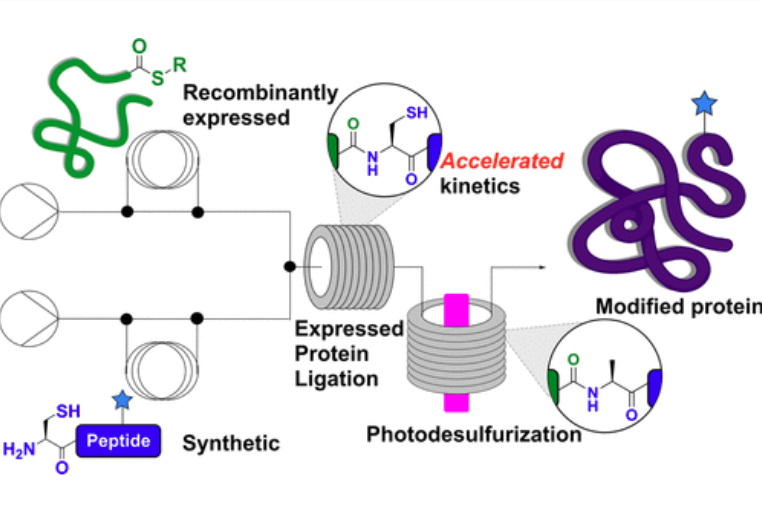
A semisynthetic extension of NCL, expressed protein ligation (EPL) [2], is an alternative transformational technology that involves the chemoselective addition of a synthetic peptide to a recombinant protein. While flow-based methods for assembly of polypeptides through NCL are well-established [3], the Payne group sought to extend the methodology and access homogenously modified semi-synthetic proteins using EPL [4] using a Vapourtec R-Series flow chemistry system, [Figure 2].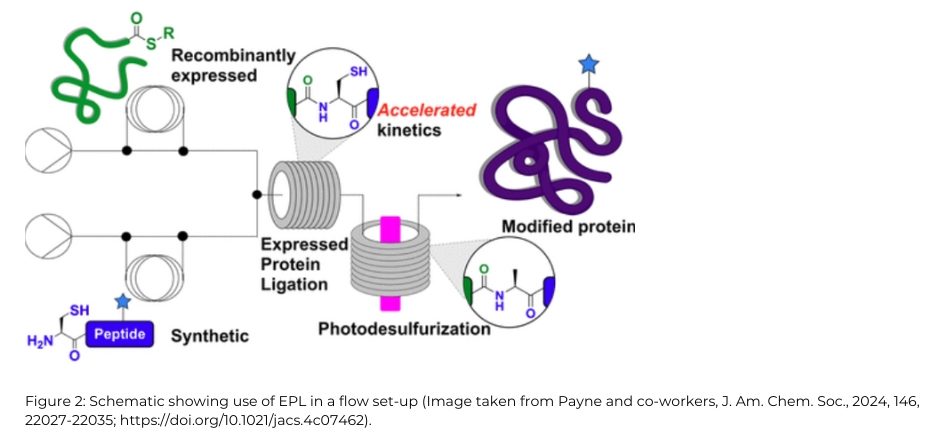
Optimisation was undertaken on the chemokine-binding evasin protein from the Cayenne tick, Amblyomma cajennense, also known as ACA-01, which contains two tyrosine sulfate modifications at both Tyr10 and Tyr12 that are essential for activity. Sulfated ACA-01 could be readily accessed in high purity by ligating a recombinantly expressed C-terminal protein fragment and a synthetic sulfopeptide thioester in flow. Once the protein had undergone folding, the semisynthetic sulfoprotein was shown to exhibit excellent binding to a range of pro-inflammatory chemokines.
A follow-up experiment prepared both unmodified and phosphorylated forms of human β-synuclein, which relied upon telescoping two processes to give an efficient, in-line flow EPL–photodesulfurization strategy. In this case it was necessary to use a demulsifying ligation buffer due to the aggregation-prone nature of the reacting protein fragment, but there was no impact upon yields or reaction rate.
In all cases:
- The flow EPL platform enabled high yields of target protein products;
- Ligation rates when using the flow set-up were vastly superior to the corresponding batch process (80 minutes in flow compared to 3 hours in batch);
- Photodesulfurization in flow was also faster (10 mins in flow compared with 1 hour in batch);
- Access to tens of milligrams of high purity material was possible.
References:
[1] Synthesis of proteins by native chemical ligation (P. E. Dawson, T. W. Muir, I. Clark-Lewis, S. B. H. Kent, Science, 1994, 266, (5186), 776−779). https://doi.org/10.1126/science.7973629
[2] Expressed protein ligation: a general method for protein engineering (T. W. Muir, D. Sondhi, P. A. Cole, Proc. Natl. Acad. Sci. U.S.A., 1998, 95 (12), 6705−6710). https://doi.org/10.1073/pnas.95.12.6705
[3] Native Chemical Ligation-Photodesulfurization in Flow (T. S. Chisholm, D. Clayton, L. J. Dowman, J. Sayers, R. J. Payne, J. Am. Chem. Soc. 2018, 140 (29), 9020−9024). https://doi.org/10.1021/jacs.8b03115
[4] Expressed Protein Ligation in Flow (L. Kambanis, A. Ayoub, M. J. Bedding, P. H. G. Egelund, J. W. C. Maxwell, C. Franck, L. Lambrechts, P. M. E. Hawkins, T. S. Chisholm, J. P. Mackay, E. Sierecki, Y. Gambin, S. S. Kulkarni, and R. J. Payne, J. Am. Chem. Soc, 2024, 146 (31), 22027-22035). https://doi.org/10.1021/jacs.4c07462