Sn-Catalyzed Criegee-Type Rearrangement of Peroxyoxindoles Enabled by Catalytic Dual Activation of Esters and Peroxides
- Moreshwar B. Chaudharia
- Krishna Jayana
- Boopathy Gnanaprakasama
- aDepartment of Chemistry, Indian Institute of Science Education and Research, Pune 411008, India
 Read the publication that featured this abstract
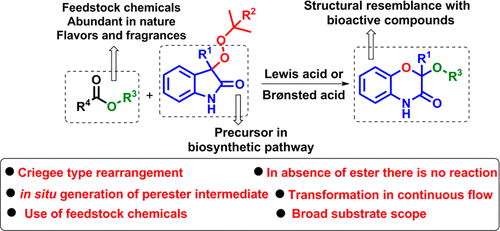
Read the publication that featured this abstractWe report here the Sn-catalyzed mild protocol for ring expansion of peroxyoxindoles to afford the series of substituted-2H-benzo[b][1,4]oxazin-3(4H)-one derivatives. In this protocol, we showed the in situ conversion of tert-butyl peroxy compounds into peresters with the aid of external esters, which then underwent the ring-expansion process, and the incipient carbocation was trapped with the alcohol residue generated from the esters. The reaction is also demonstrated in a continuous flow process to afford the rearranged product in 22 min of residence time.
Get in touch
For more information on flow chemistry systems and services please use the contact methods below.
Call us on +44 (0)1284 728659 or Email us
Resource Centre
R-Series

The Vapourtec R-Series is, quite simply, unrivalled for flow chemistry
- Flexible |
- Precise |
- Automatable
The R-Series is undoubtedly the most versatile, modular flow chemistry system available today.
E-Series

The Vapourtec E-Series is the perfect introductory system for flow chemistry
- Robust |
- Easy to use |
- Affordable
The E-Series is a robust and affordable, entry level flow chemistry system designed for reliability and ease of use.
