Development of a palladium-catalyzed decarboxylative arene cross-coupling of pyrrole derivatives in a flow reactor
- Cindy Buonomanoa,b
- Michael Holtz-Mulhollanda,b
- Sydney Sullivana,b
- Pat Forgionea,b
- aDepartment of Chemistry and Biochemistry, Concordia University, 7141 Sherbrooke O, Montréal, QC, H4B 1R6, Canada
- bCentre in Green Chemistry and Catalysis, Montréal, QC, H3C 3J7, Canada
 Read the publication that featured this abstract
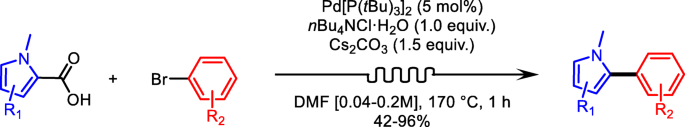
Read the publication that featured this abstractPalladium-catalyzed decarboxylative cross-coupling was employed to synthesize 2-arylpyrroles via a flow process. This reaction features palladium as the only metal catalyst and uses easily accessible starting materials. The reaction temperature, the residence time, and the quantity of different reactants were investigated to achieve optimal reaction conditions. A variety of N-alkylated and N-arylated 2-arylpyrroles were produced in good to excellent yields. A N-methyl-2-arylpyrrole derivative was produced in 220 min on a 3 g scale in 84% yield. The flow set-up presented in this work is featuring a fixed bed reactor to load the insoluble Cs2CO3 necessary for the decarboxylative cross-coupling to occur, it also comprises a sample loop, and a stainless-steel reactor. This study demonstrated the excellent potential of utilizing a flow process for the synthesis of 2-arylpyrroles derivatives.
Get in touch
For more information on flow chemistry systems and services please use the contact methods below.
Call us on +44 (0)1284 728659 or Email us
Resource Centre
R-Series

The Vapourtec R-Series is, quite simply, unrivalled for flow chemistry
- Flexible |
- Precise |
- Automatable
The R-Series is undoubtedly the most versatile, modular flow chemistry system available today.
E-Series

The Vapourtec E-Series is the perfect introductory system for flow chemistry
- Robust |
- Easy to use |
- Affordable
The E-Series is a robust and affordable, entry level flow chemistry system designed for reliability and ease of use.
