Application of Flow and Biocatalytic Transaminase Technology for the Synthesis of a 1-Oxa-8-azaspiro [4.5] decan-3-amine
- Jeffrey T. Kohrta
- Peter H. Dorffa
- Michael Burnsb
- Chewah Leeb
- Steven V. O’Neilb
- Robert J. Maguireb
- Rajesh Kumarb
- Melissa Wagenaarb
- Loren Pricea
- Manjinder S. Lalla
- aMedicine Design, Pfizer Inc., 445 Eastern Point Road, Groton, Connecticut 06340, United States
- bChemical Research and Development, Pfizer Inc., 445 Eastern Point Road, Groton, Connecticut 06340, United States
 Read the publication that featured this abstract
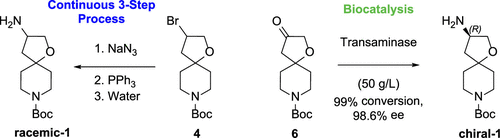
Read the publication that featured this abstractSpirocyclic ring systems are useful intermediates in the design and synthesis of medicinally active agents and commonly found as cores in natural products. Recently, syntheses of a key intermediate Boc-protected-1-oxa-8-azaspiro[4.5]decan-3-amine 1 were examined. While multigram quantities of the racemic material could be made from the reduction of an energic azide intermediate, larger scale reactions and a chiral synthesis required further investigations. Herein, we describe the use of a continuous three-step flow process to scale the formation and reduction of an azide intermediate, and the use of a transaminase to prepare the desired enantiomer in high yield and enantiomeric excess.
Get in touch
For more information on flow chemistry systems and services please use the contact methods below.
Call us on +44 (0)1284 728659 or Email us
Resource Centre
R-Series

The Vapourtec R-Series is, quite simply, unrivalled for flow chemistry
- Flexible |
- Precise |
- Automatable
The R-Series is undoubtedly the most versatile, modular flow chemistry system available today.
E-Series

The Vapourtec E-Series is the perfect introductory system for flow chemistry
- Robust |
- Easy to use |
- Affordable
The E-Series is a robust and affordable, entry level flow chemistry system designed for reliability and ease of use.
