Continuous-flow generation of diazoesters and their direct use in S-H and P-H insertion reactions: synthesis of a-sulfanyl, a-sulfonyl and a-phosphono carboxylates
- Hannah E. Bartrum1
- David C. Blakemore2
- Christopher J. Moody1
- Christopher J. Hayes1
- 1School of Chemistry, University of Nottingham, University Park, Nottingham, NG7 2RD, UK
- 2Pfizer Neusentis, The Portway Building, Granta Park, Cambridge, CB21 6GS, UK
 Read the publication that featured this abstract
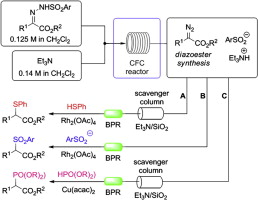
Read the publication that featured this abstractThe synthesis of α-sulfanyl, α-sulfonyl, and α-phosphono carboxylates has been achieved using a two-step procedure involving the in-flow generation of diazoesters from sulfonylhydrazones, via Bamford–Stevens elimination, and then subsequent S–H, sulfinate, and P–H carbene insertion reactions. The method for α-sulfonyl ester is particularly noteworthy as it represents a very atom economic (‘green’) way to access the products, and it completely avoids the use of alkyl halides.
Get in touch
For more information on flow chemistry systems and services please use the contact methods below.
Call us on +44 (0)1284 728659 or Email us
Resource Centre
R-Series

The Vapourtec R-Series is, quite simply, unrivalled for flow chemistry
- Flexible |
- Precise |
- Automatable
The R-Series is undoubtedly the most versatile, modular flow chemistry system available today.
E-Series

The Vapourtec E-Series is the perfect introductory system for flow chemistry
- Robust |
- Easy to use |
- Affordable
The E-Series is a robust and affordable, entry level flow chemistry system designed for reliability and ease of use.
